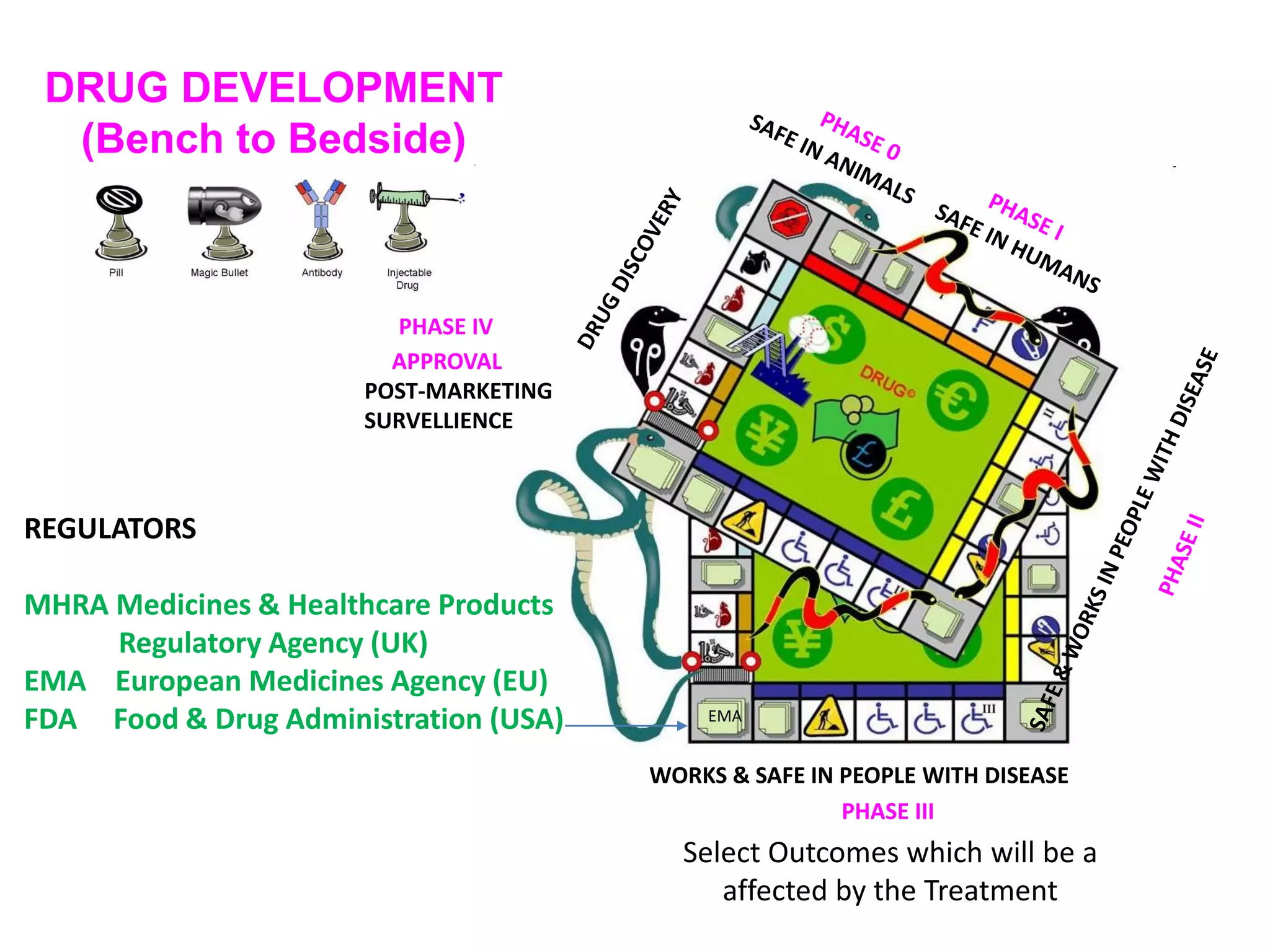
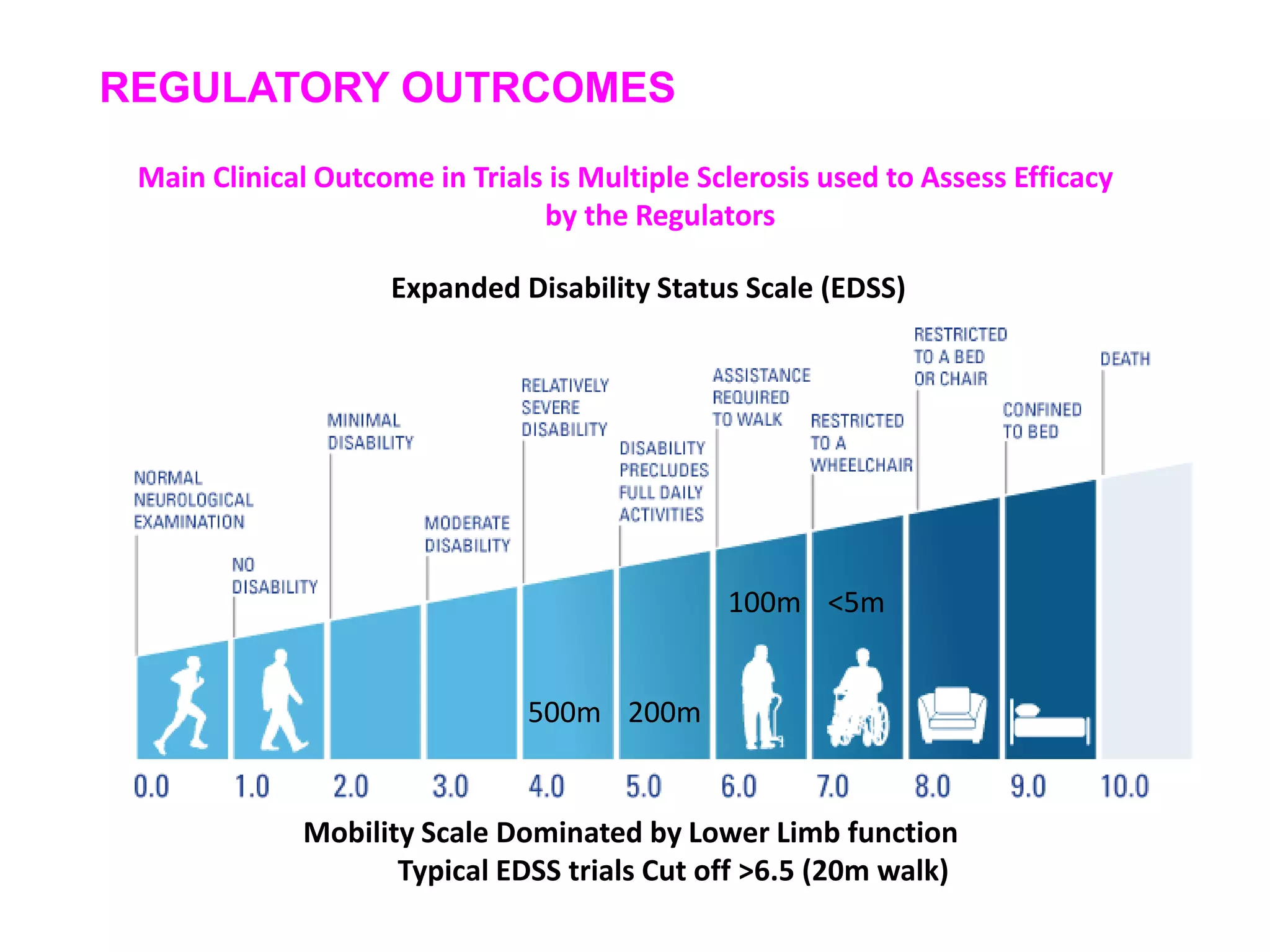
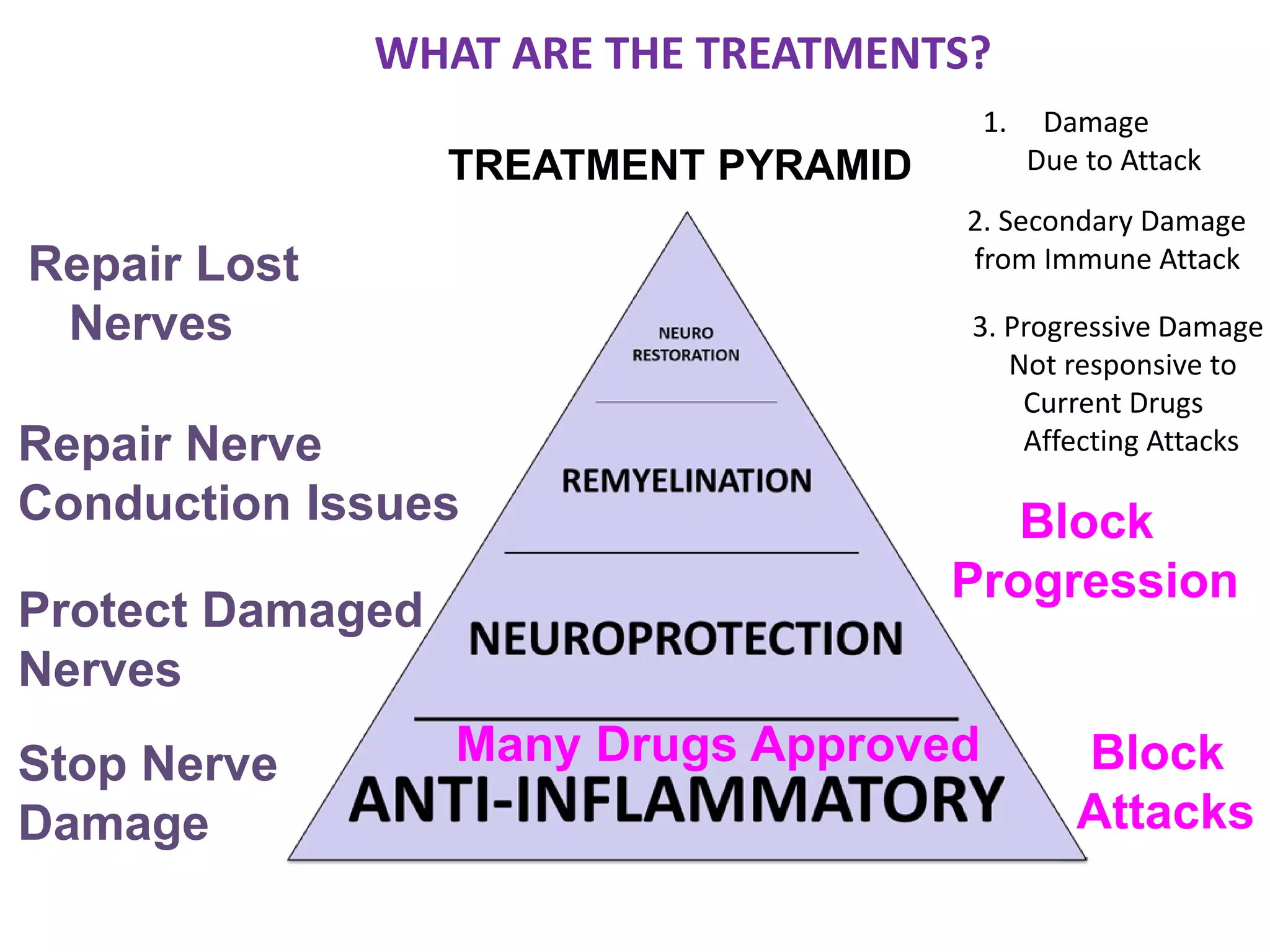
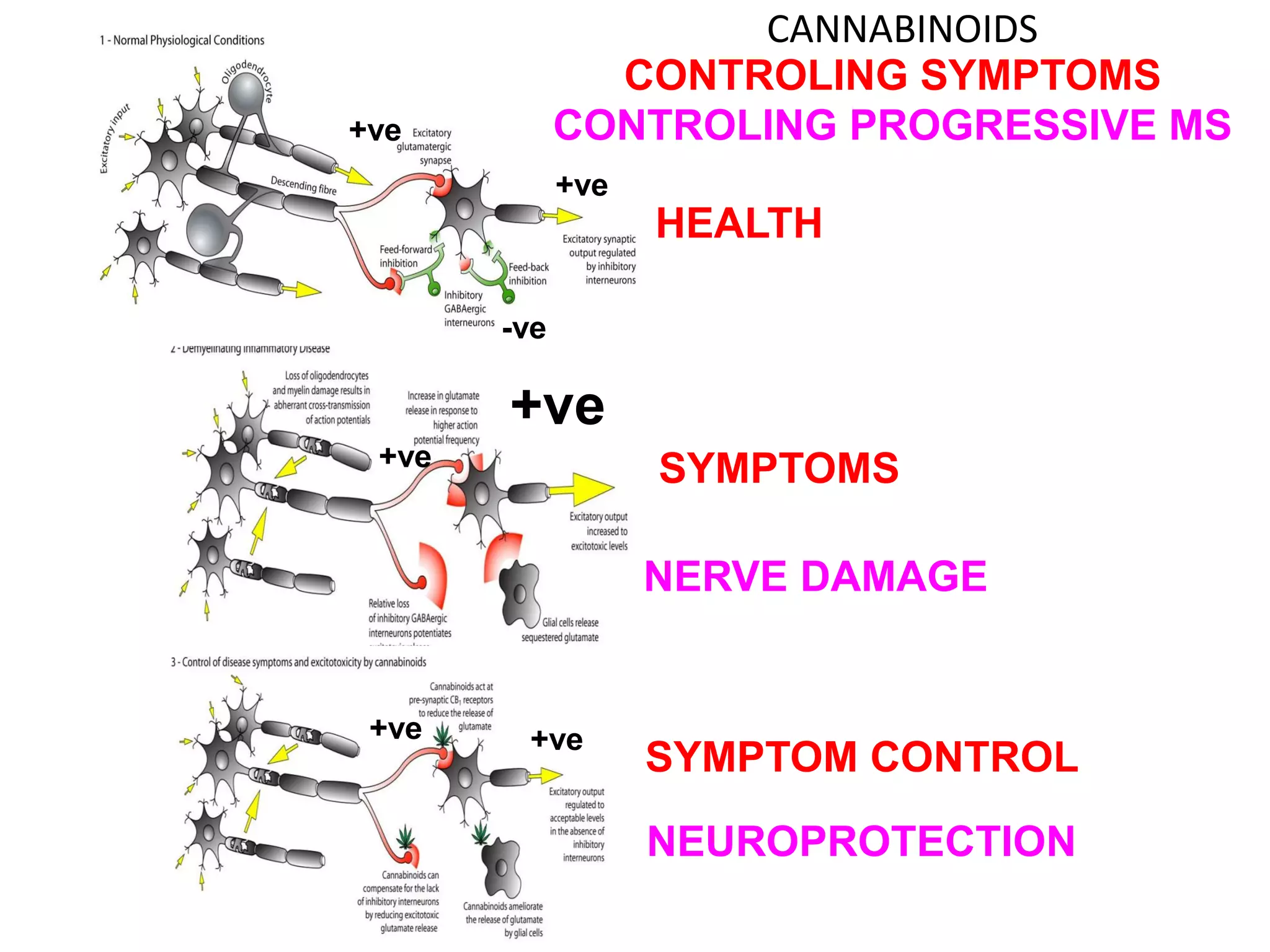
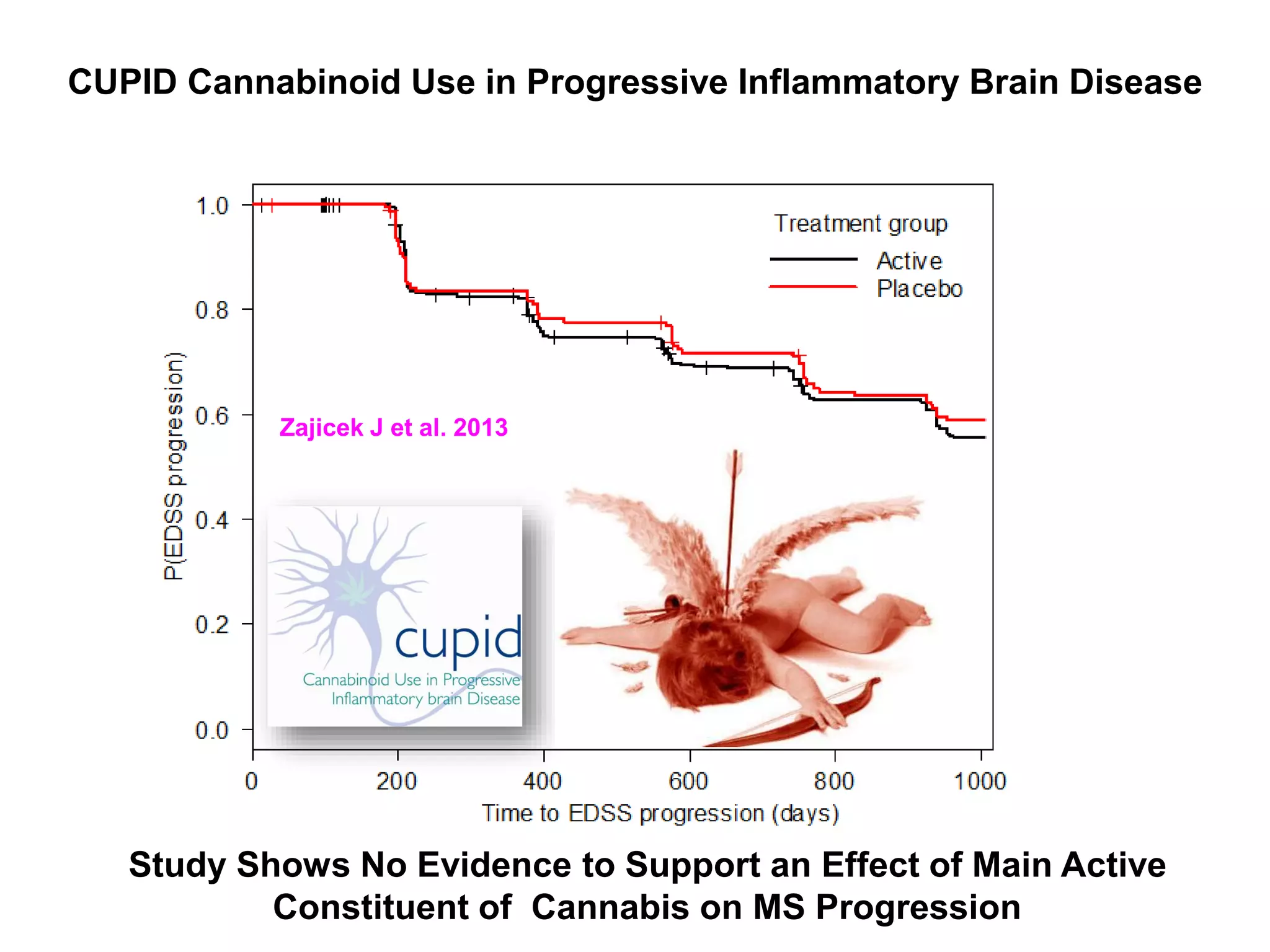
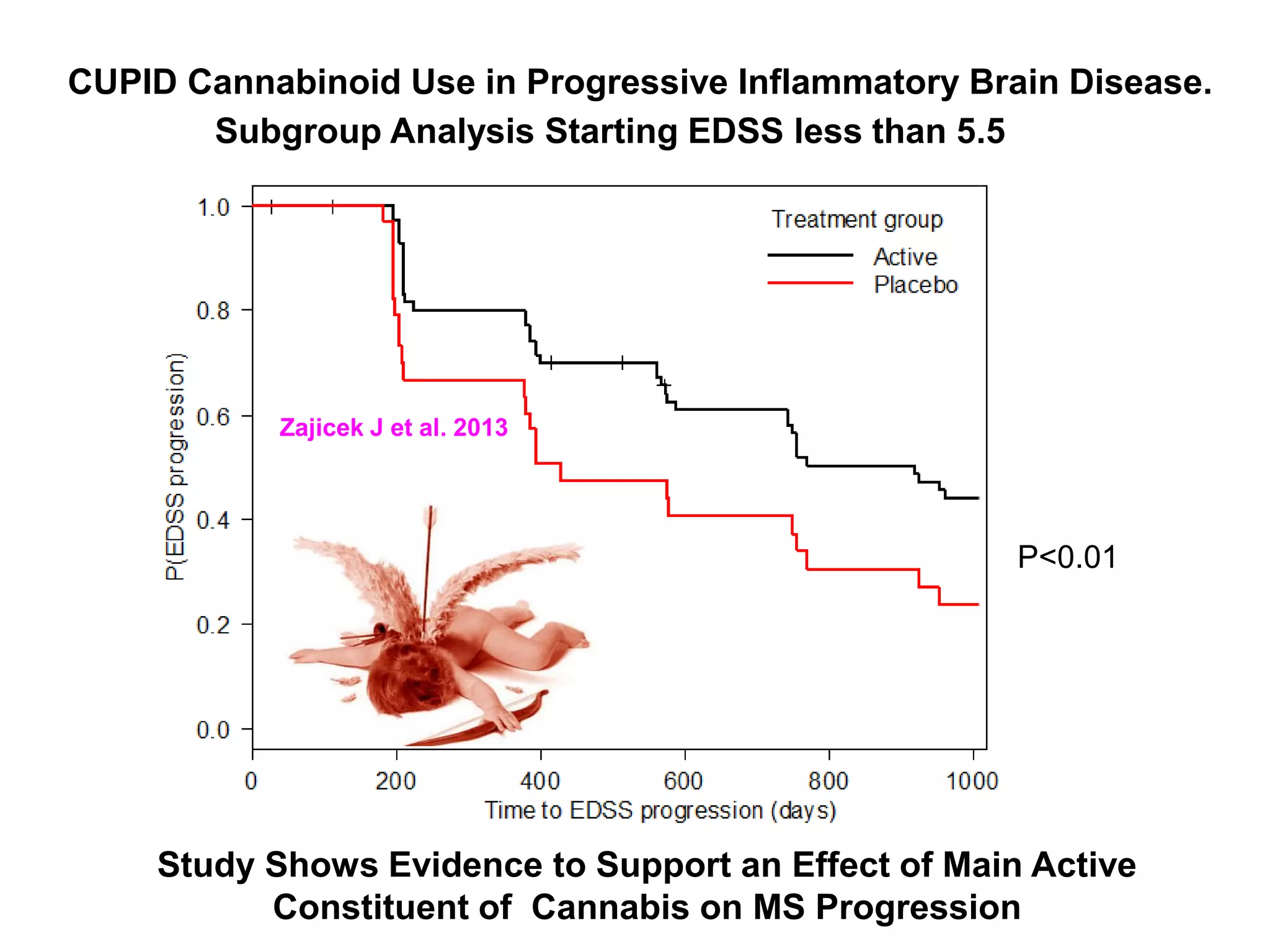
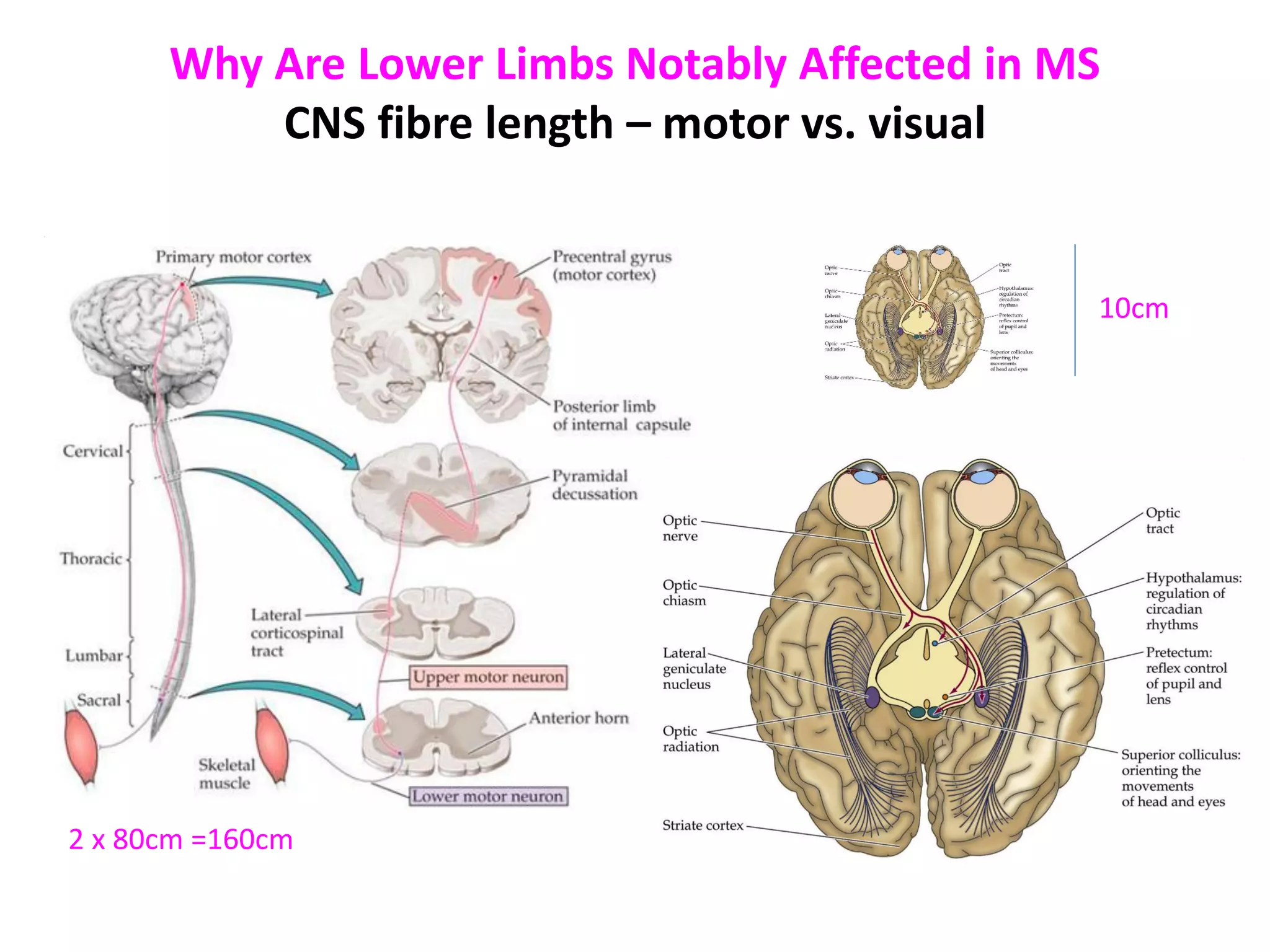
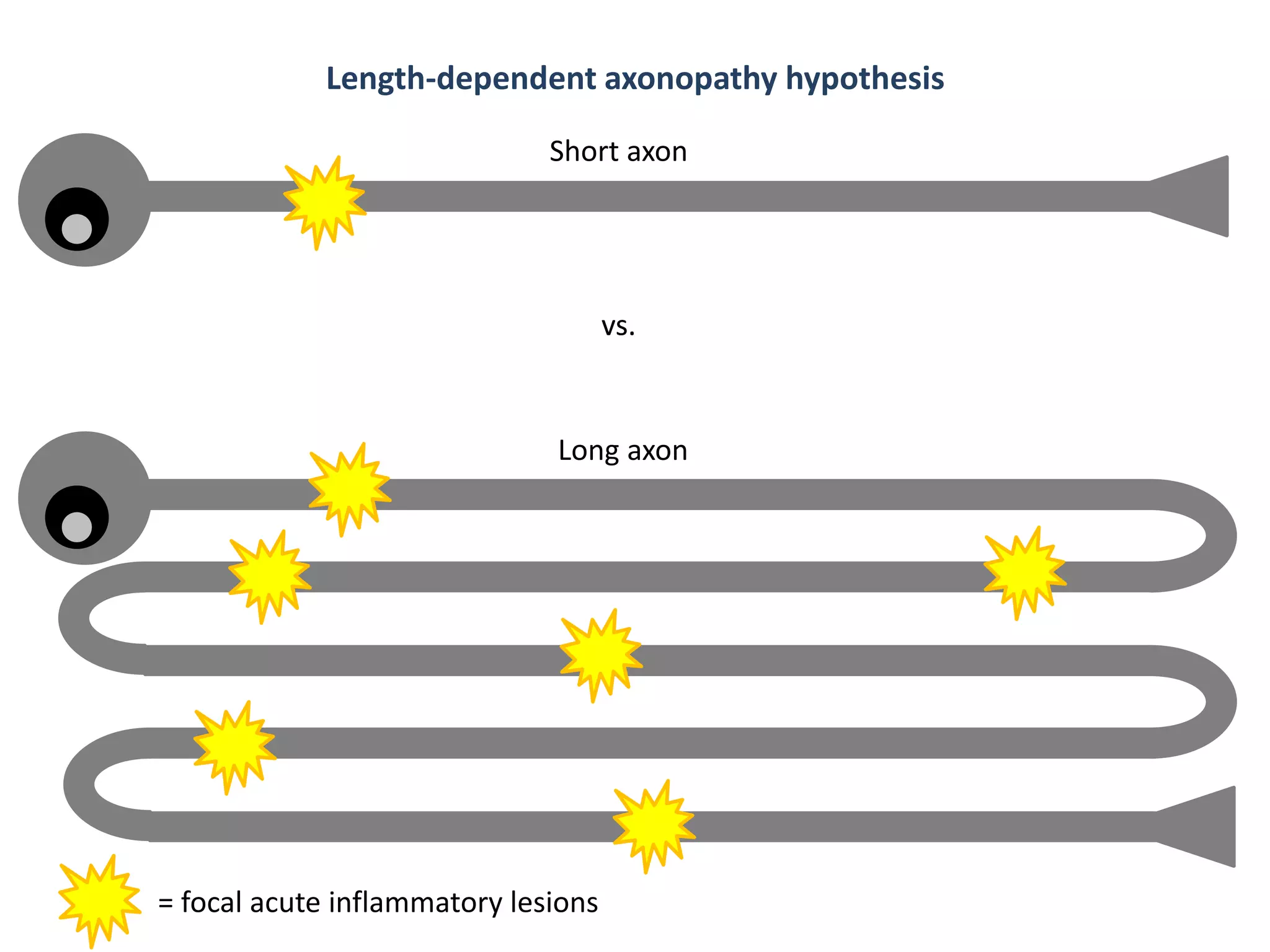
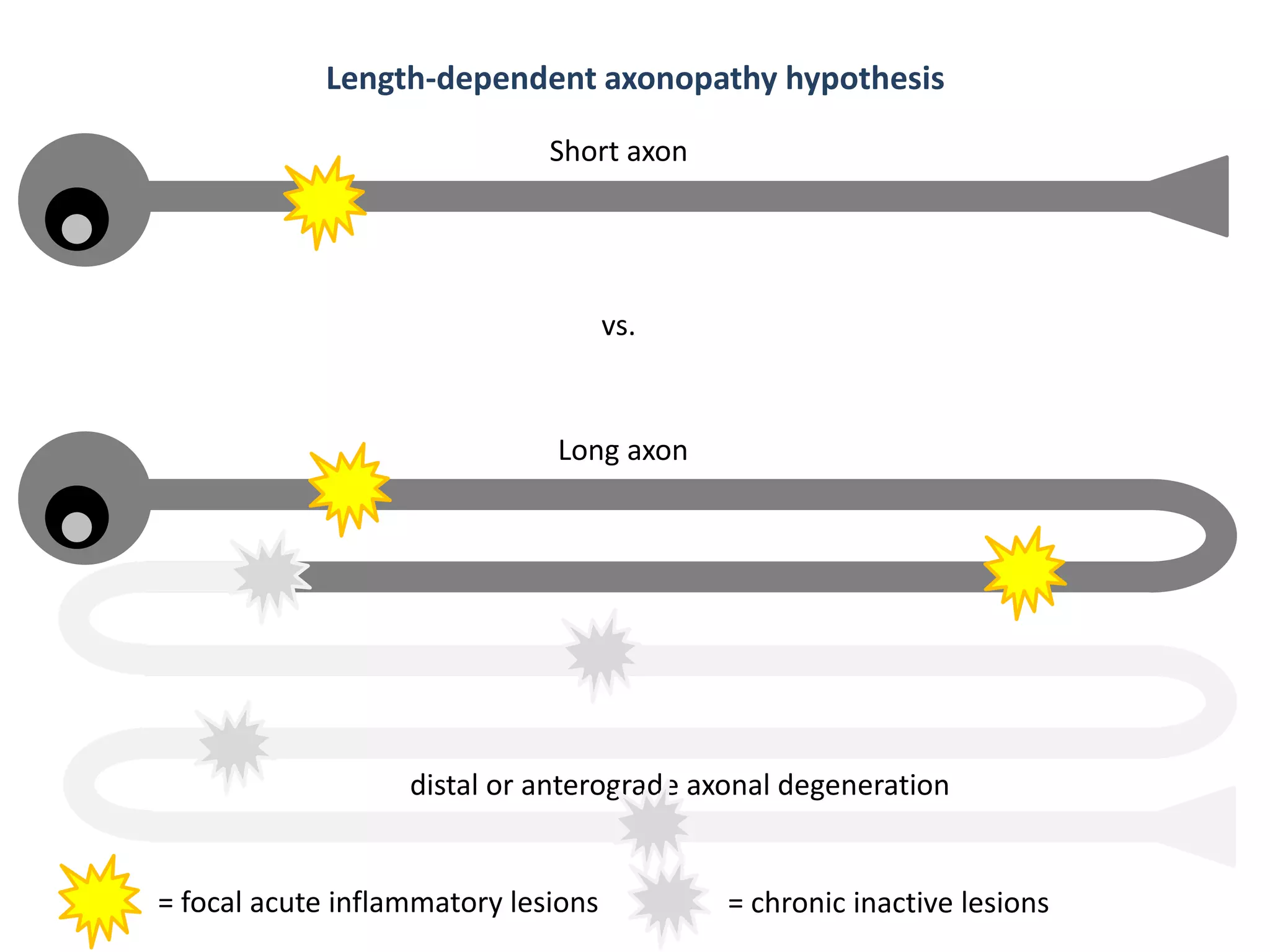
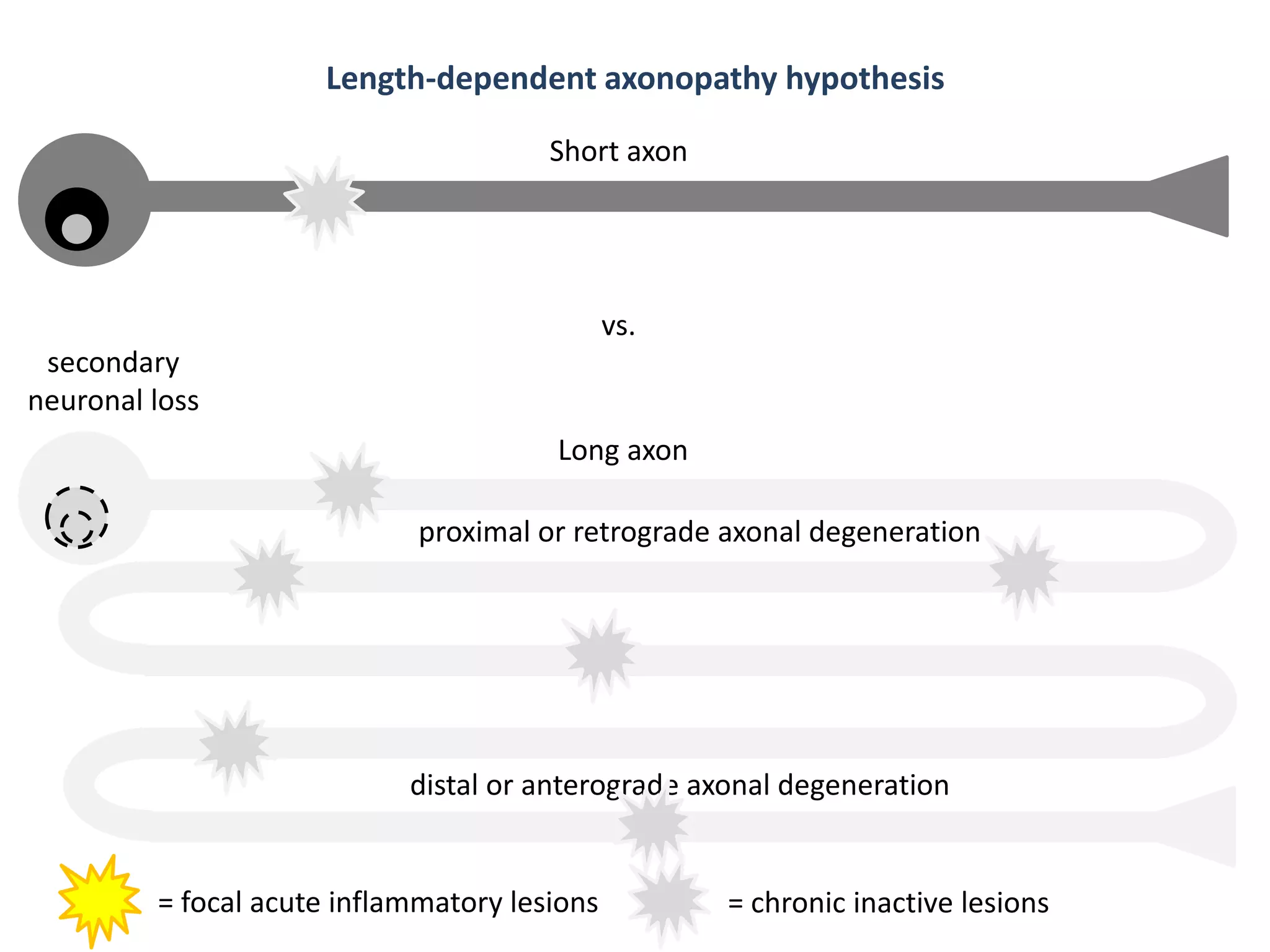
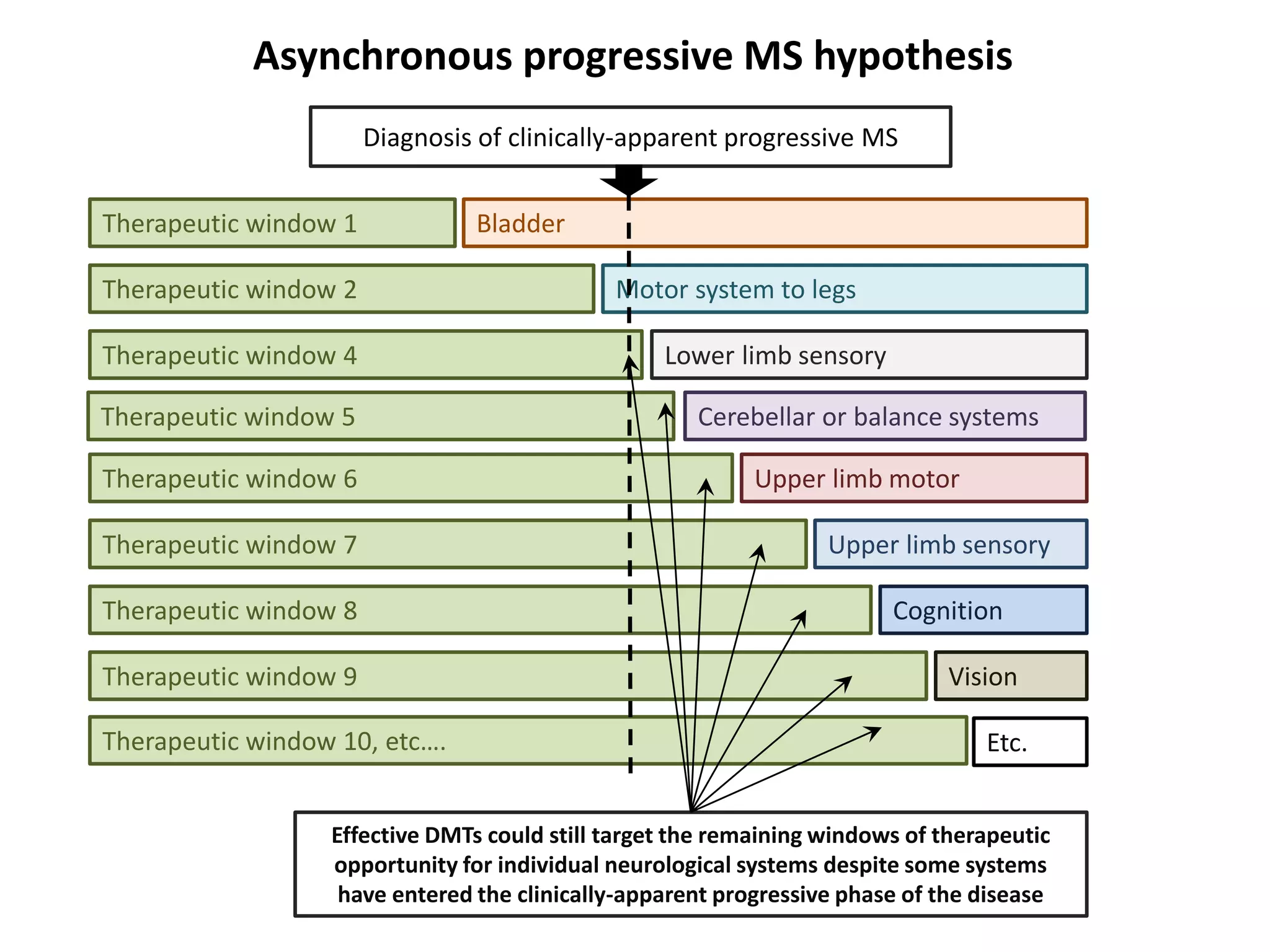
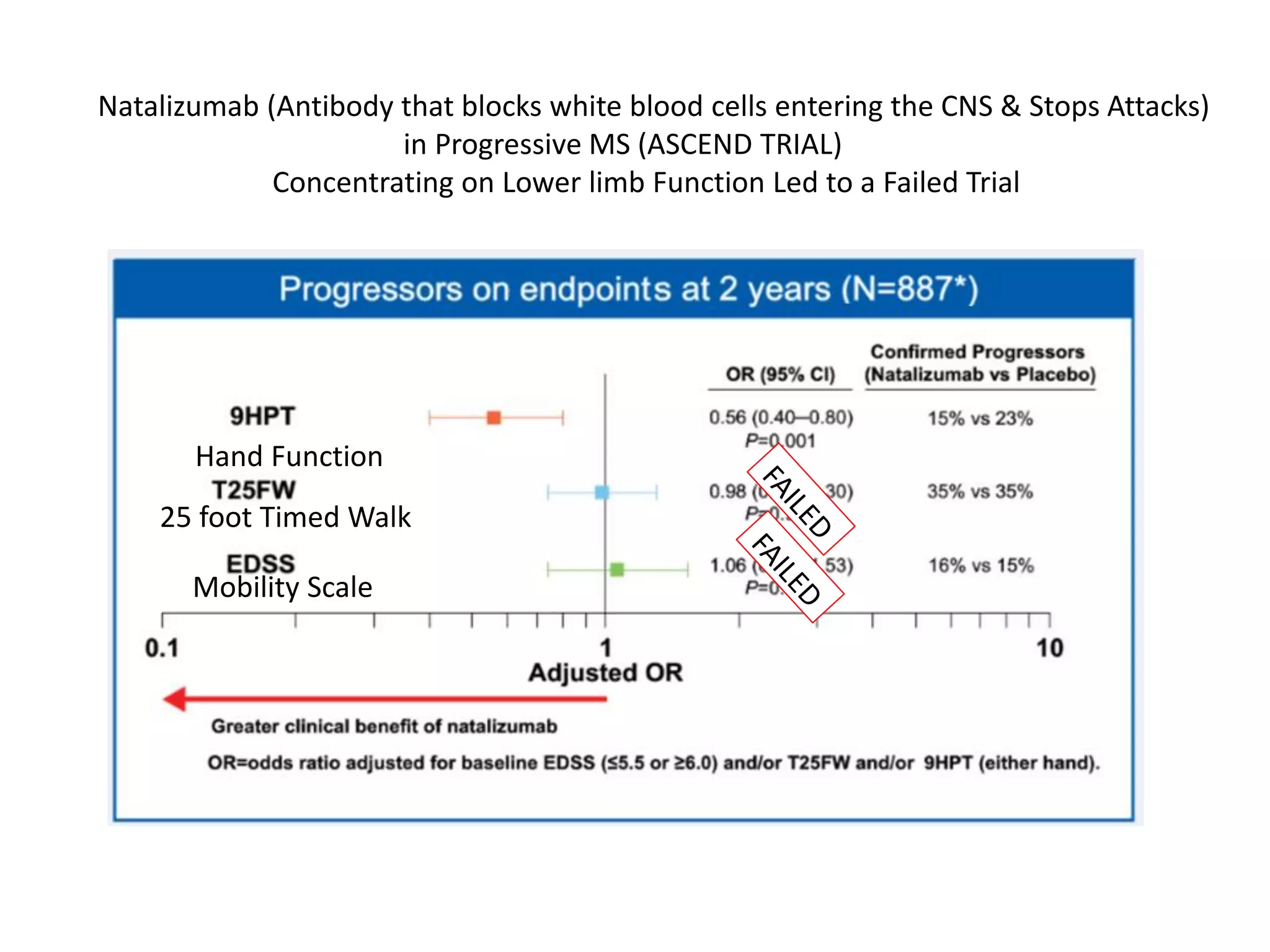
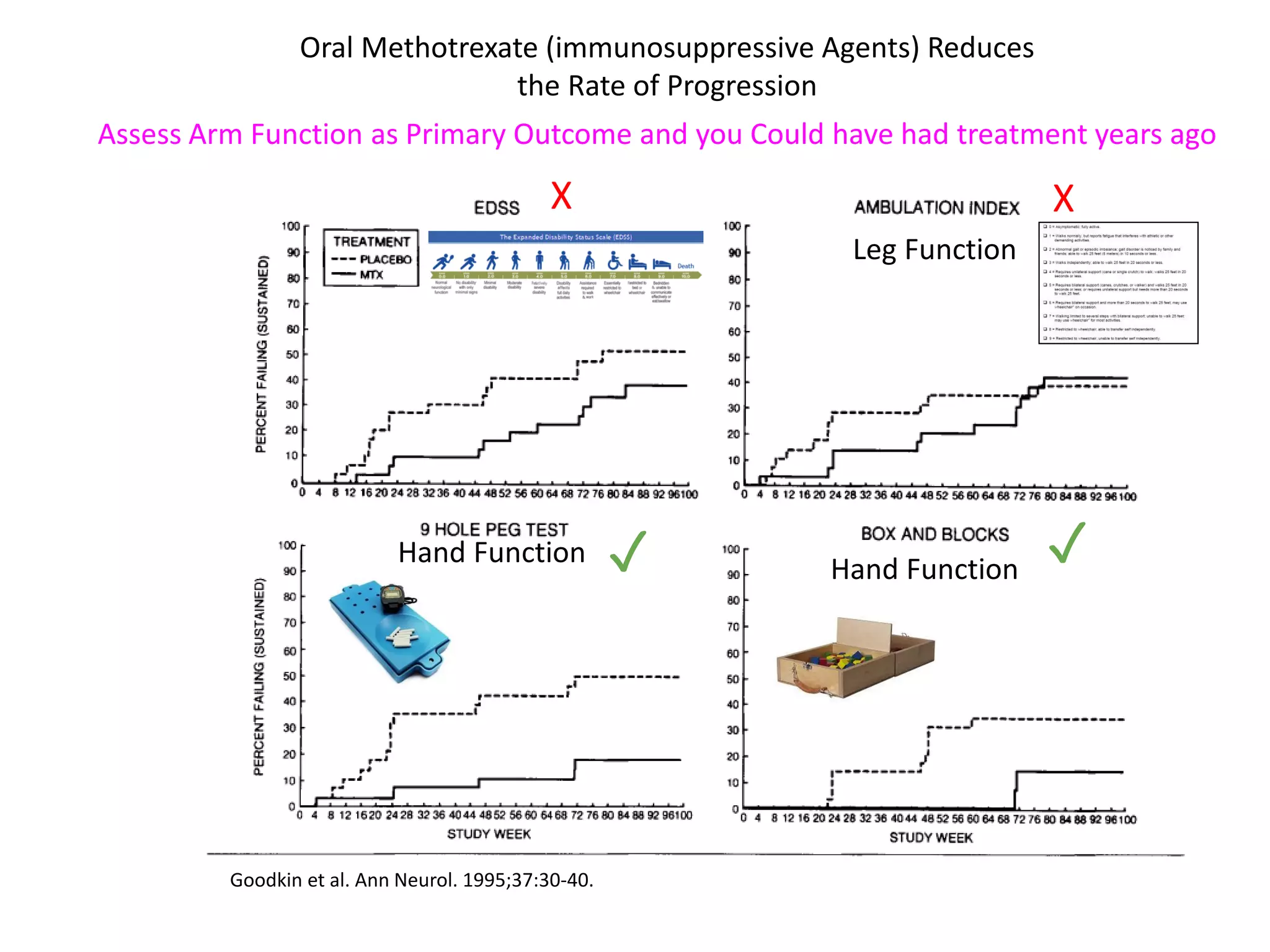
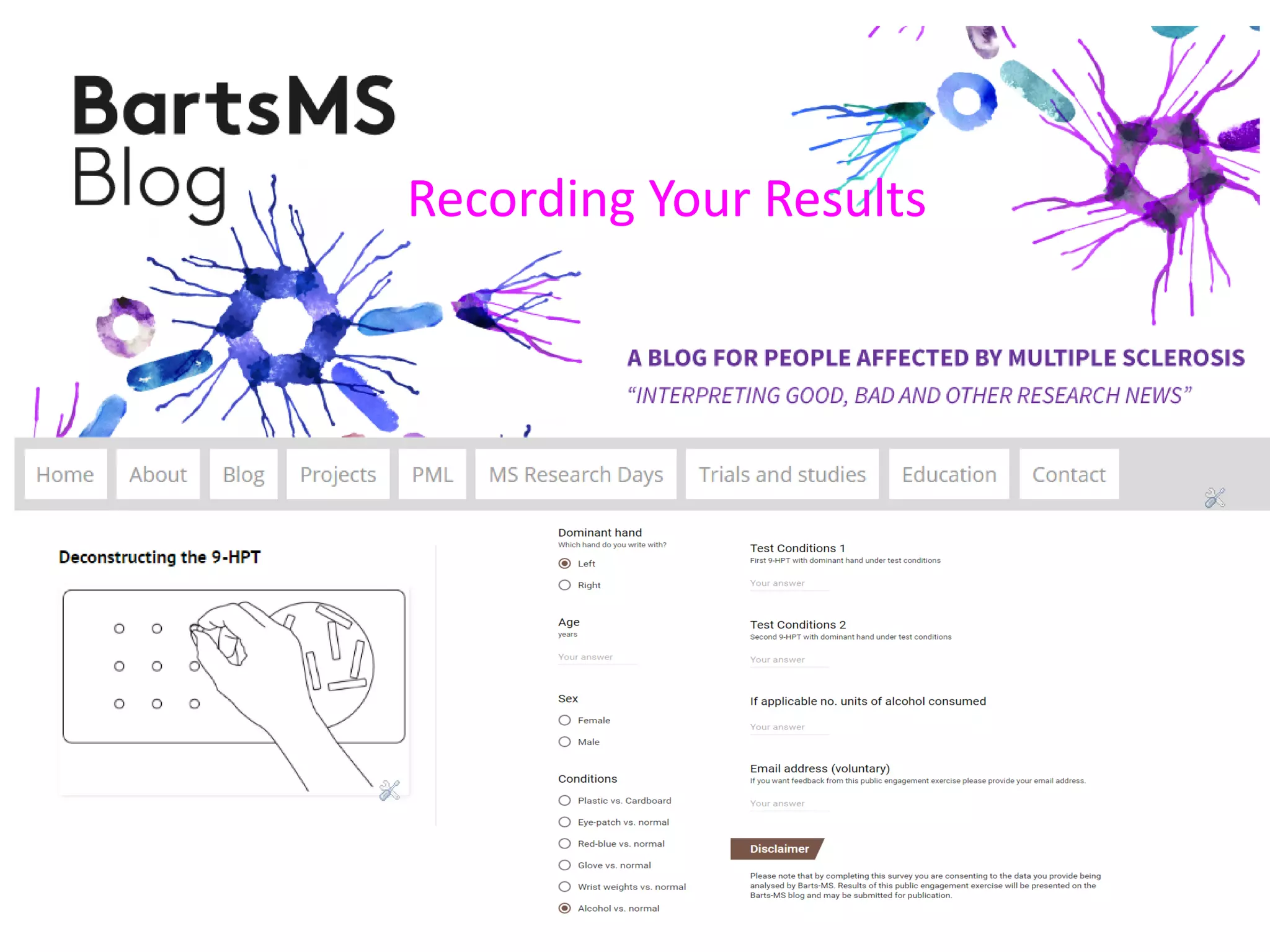
This document discusses the importance of hand function in assessing treatment outcomes for multiple sclerosis (MS). It notes that current clinical trials and regulatory outcomes primarily focus on mobility scales that are dominated by lower limb function, excluding people who use wheelchairs. It argues that assessing hand function could have led to different outcomes in past trials and enabled treatments years ago. The document advocates increasing awareness of hand function for people with MS, neurologists, and regulators. It promotes developing new patient-reported outcome measures that better capture elements of hand function that are important to people living with MS.