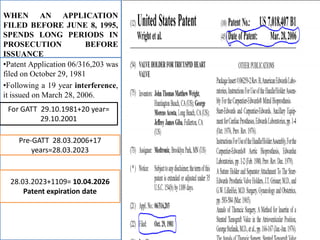
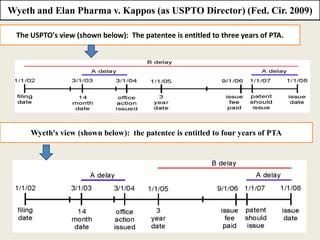
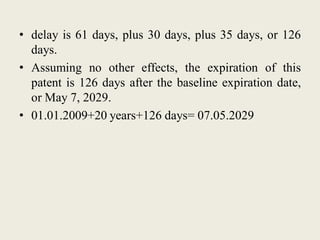Patent Term Adjustment (PTA) provides additional time to a patent term to compensate patent holders for certain delays introduced by the USPTO during patent prosecution. PTA is calculated based on three types of delays: A) USPTO delays in responding to filings; B) delays when prosecution exceeds three years; and C) delays due to interferences, appeals, or secrecy orders. The amount of PTA is determined by adding together delays from these types, subtracting overlapping delays or applicant-caused delays, and cannot exceed five years. Two important court cases, Wyeth v. Kappos and Novartis v. Lee, impacted PTA calculations by clarifying how the USPTO should determine








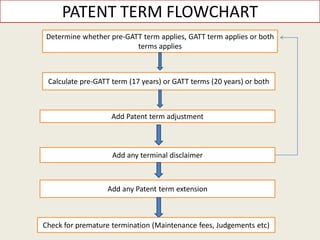
![“Baseline” Patent Term Pre-GATT or GATT (17 or 20 years)
• The baseline term of a utility patent (or plant patent) is either the pre-GATT 17 year term, or
the GATT 20 year term.
• The determination of which baseline term applies is governed by 35 U.S.C. § 154(c)(1): The
term of a patent that is in force on or that results from an application filed before the date
that is 6 months after the date of the enactment of the Uruguay Round Agreements Act [i.e.,
June 8, 1995] shall be the greater of the 20 year term as provided in subsection (a) [i.e., the
GATT term], or 17 years from grant, subject to any terminal disclaimers.](https://image.slidesharecdn.com/6f941bcb-df84-4014-8a03-3a6cf91be4bd-160613165911/85/PRESENTATION-ON-PATENT-TERM-ADJUSTMENT-PTA-10-320.jpg)