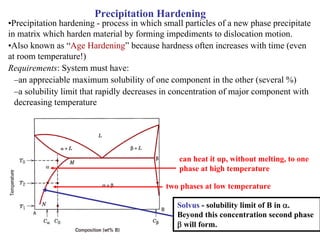
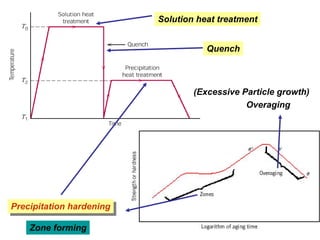
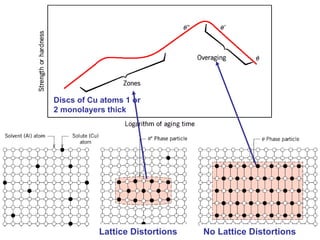
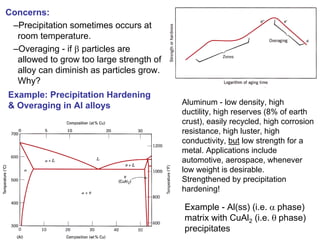
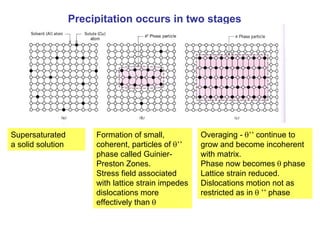
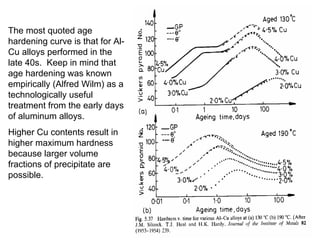
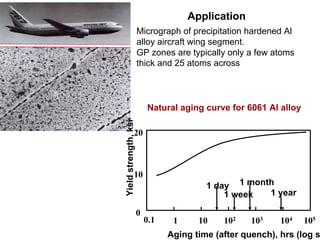
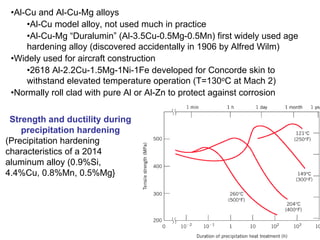
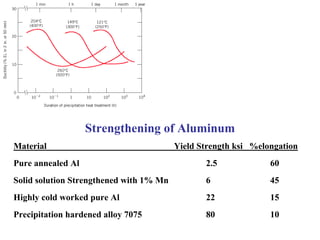
Dispersion strengthening and precipitation hardening can significantly increase the strength of metal alloys through the addition of uniformly distributed particles within the metal matrix. During precipitation hardening, heating and quenching treatments cause particles to precipitate out of solid solution, impeding dislocation motion and strengthening the material. The size, amount, and distribution of precipitate particles controls the resulting mechanical properties of the alloy. Common alloys strengthened this way include aluminum, nickel, titanium, and iron-based alloys.