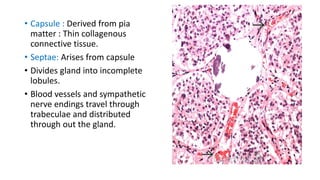
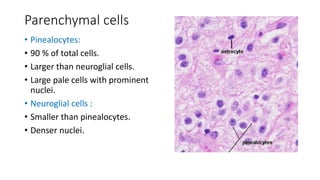
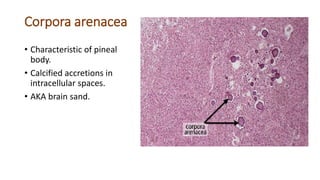
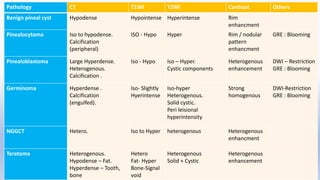
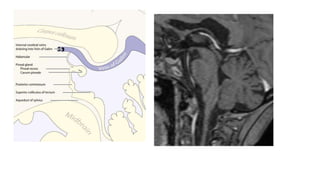
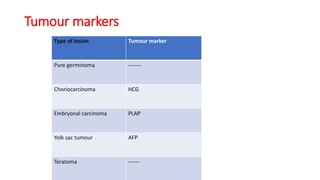
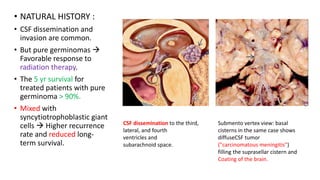
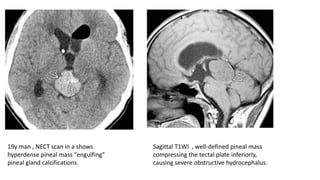
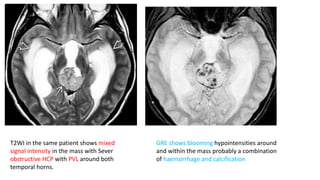
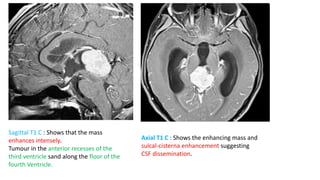
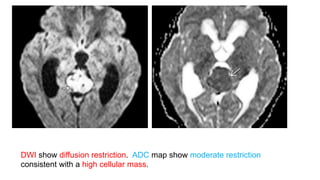
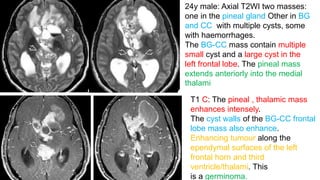
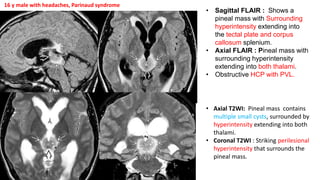
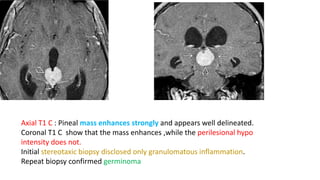
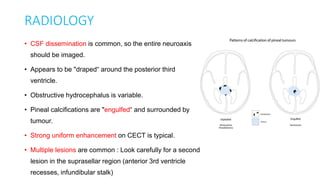
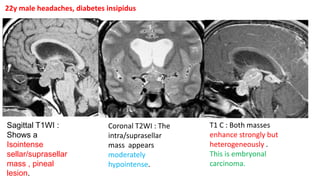
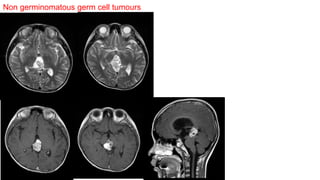
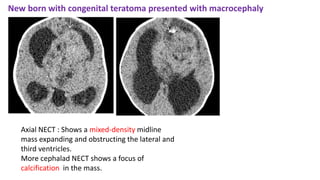
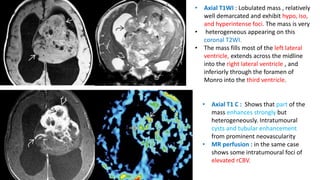
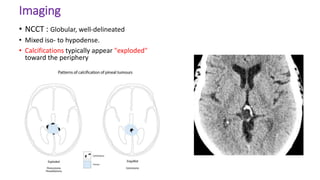
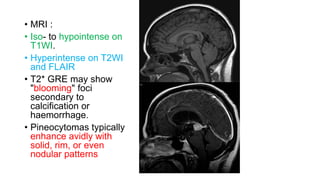
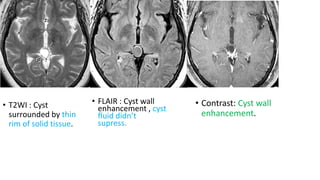
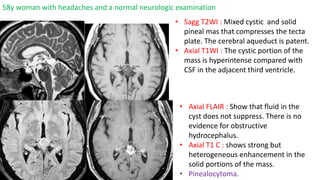
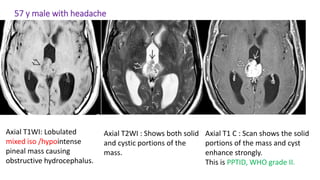
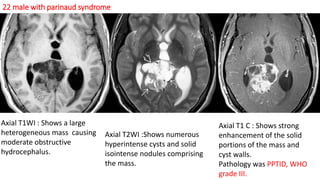
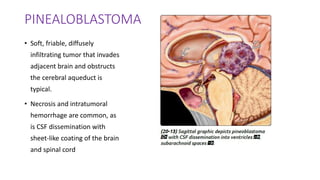
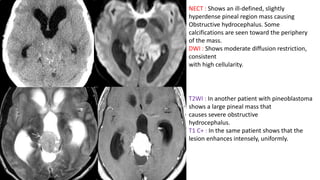
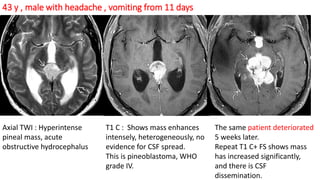
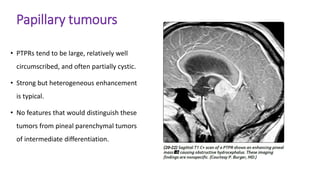
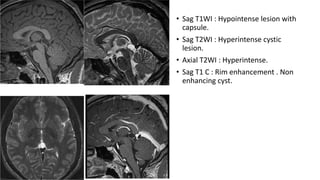
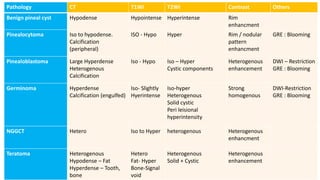
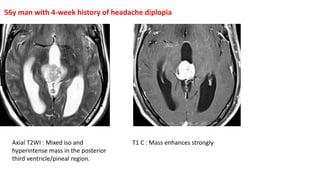
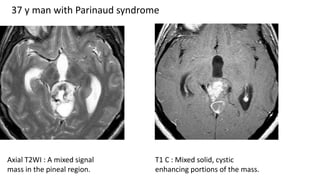
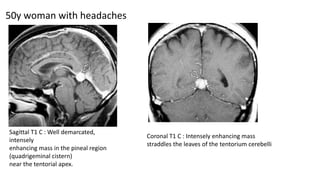
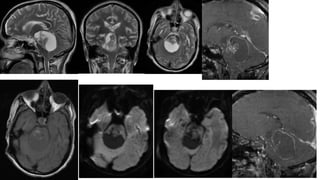
This document provides an overview of pineal region tumors, including their clinical features, radiology, and histology. It discusses the main tumor types seen in the pineal region, including germ cell tumors, pineal parenchymal cell tumors, and glial cell tumors. Germ cell tumors are the most common pineal region tumors in children and young adults. Clinical features vary depending on the tumor location and can include increased intracranial pressure, cranial nerve deficits, and endocrine dysfunction. Radiologically, pineal region tumors often present as enhancing masses that may engulf or displace the pineal gland. Specific tumor types have characteristic imaging patterns described in the document.