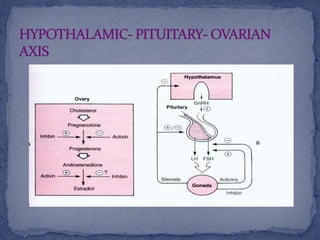
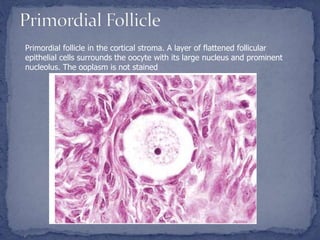
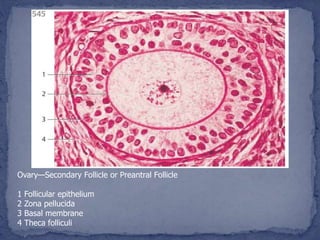
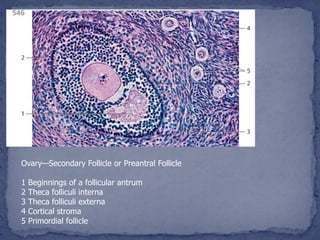
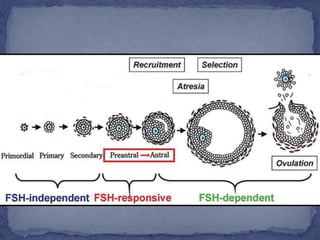
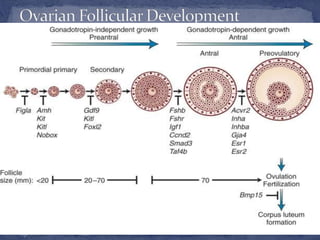
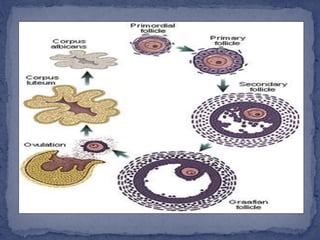
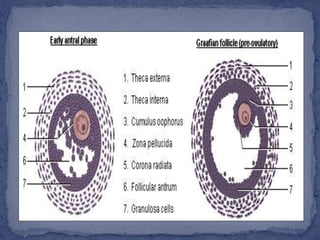
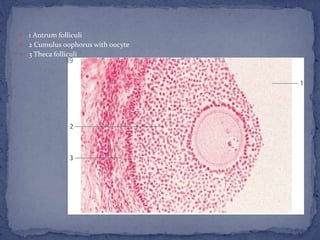
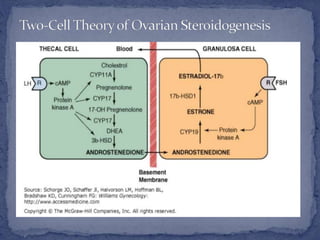
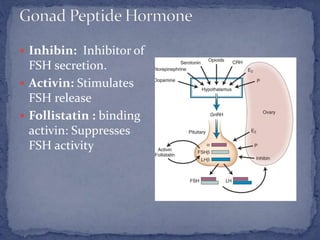
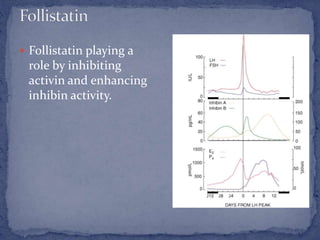
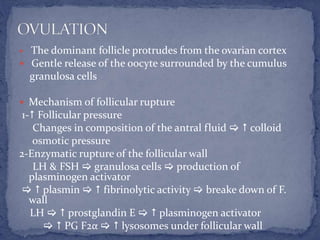
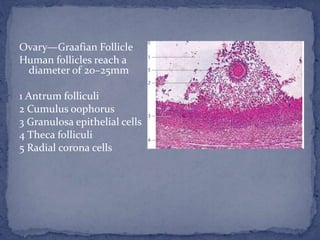
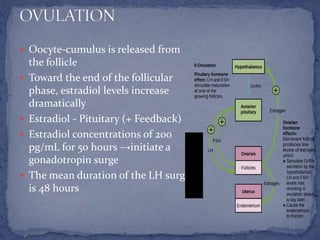
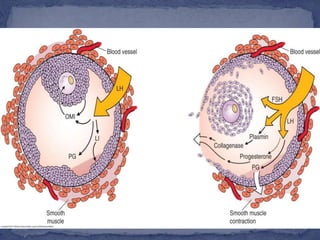
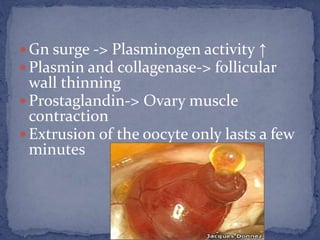
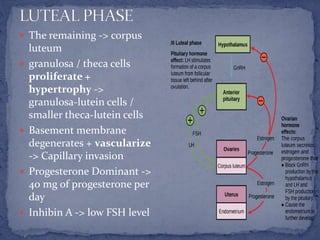
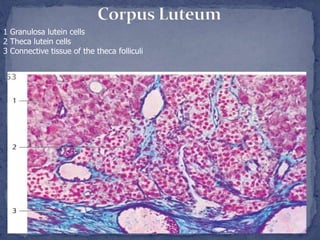
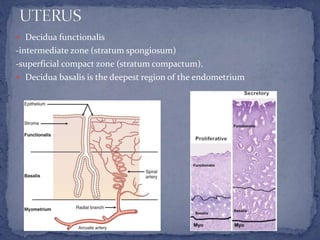
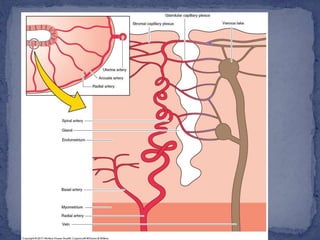
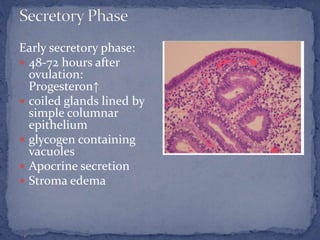
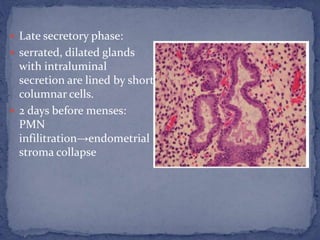
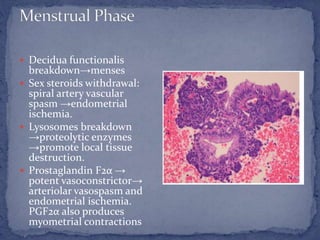
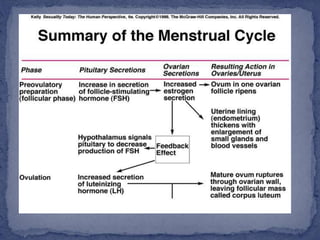
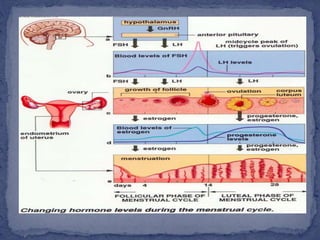
The menstrual cycle is regulated by the hypothalamus, pituitary gland, and ovaries. It typically lasts 28 days and is divided into the follicular phase and luteal phase. During the follicular phase, follicle stimulating hormone (FSH) causes follicles to mature in the ovaries with one becoming dominant. Around day 14, luteinizing hormone (LH) surges causing ovulation of the dominant follicle. The remaining follicle cells form the corpus luteum which secretes progesterone to prepare the endometrium for potential implantation. If implantation does not occur, progesterone and estrogen levels fall, causing menstruation and the start of a new cycle.