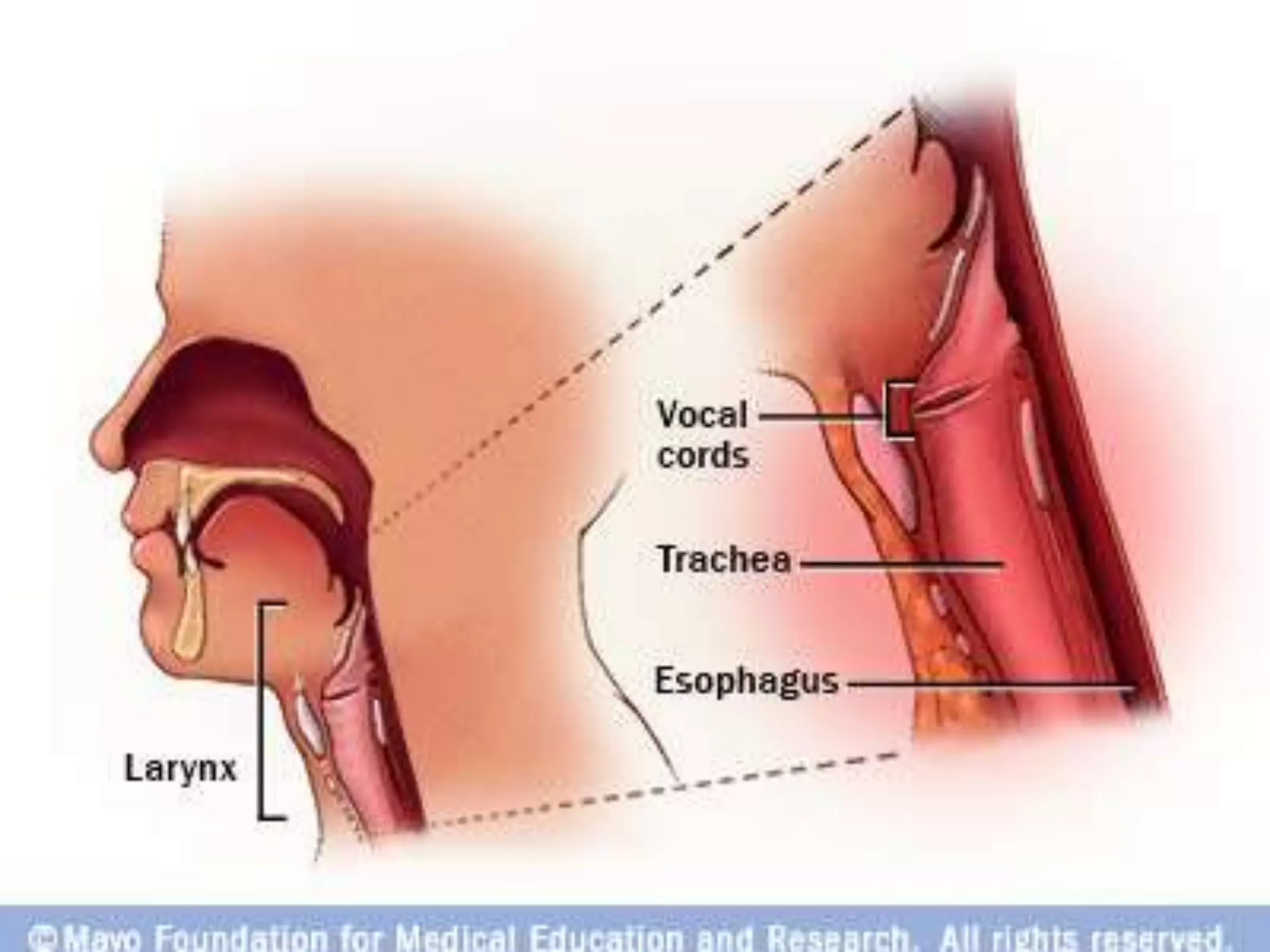
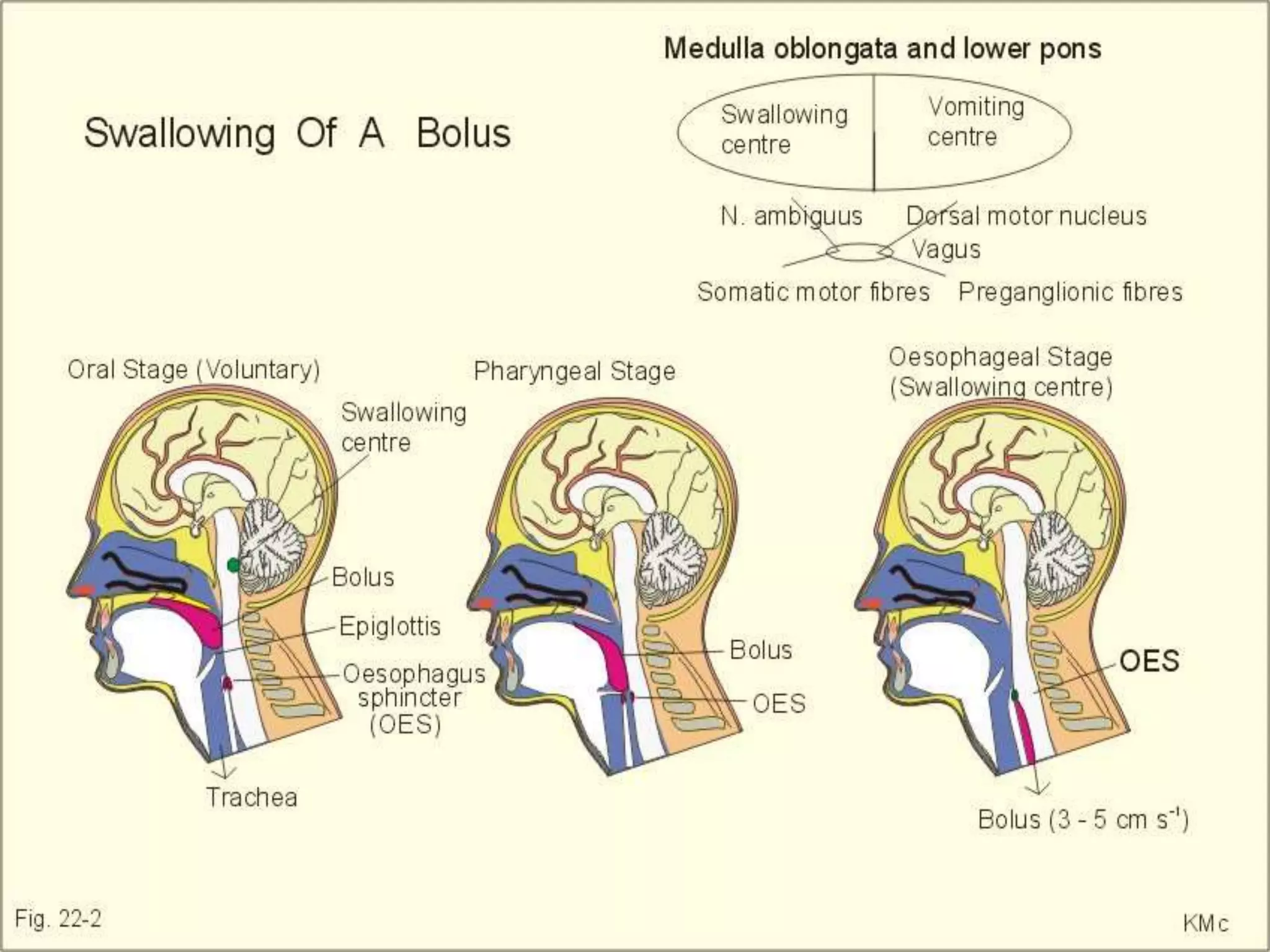
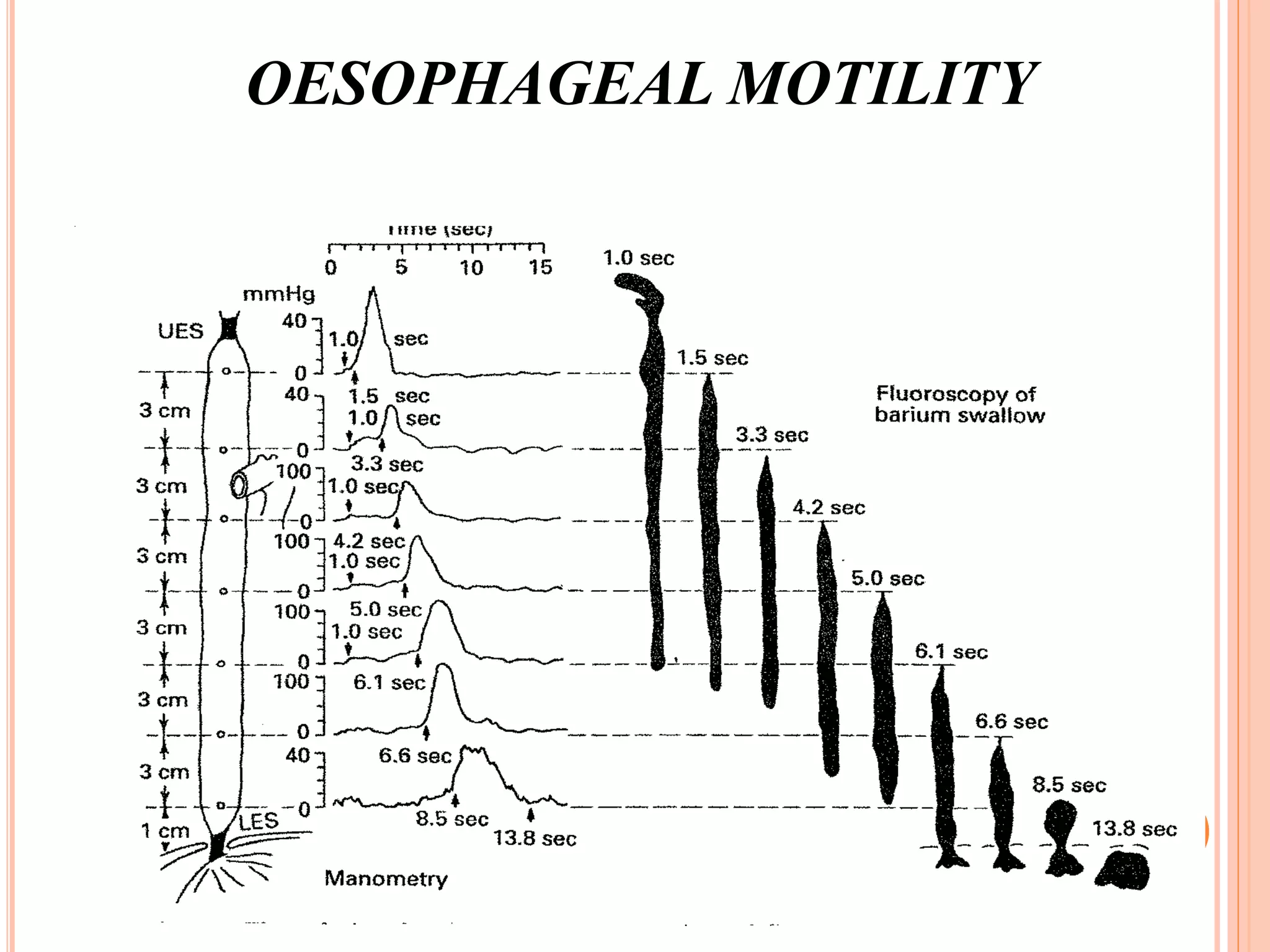
This document provides an overview of deglutition (swallowing) physiology and esophageal manometry. It describes the three phases of swallowing (oral, pharyngeal, esophageal) and the muscles involved in each phase. It also outlines the neural control of swallowing and the brainstem nuclei involved. Regarding esophageal manometry, it describes the different catheter types, indications for the test, and provides a detailed outline of the components and steps to perform esophageal manometry including identifying high pressure zones and measuring lower esophageal sphincter relaxation.