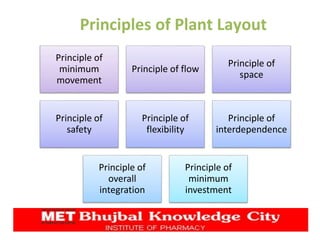
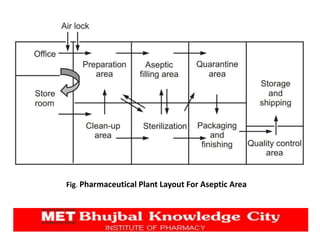
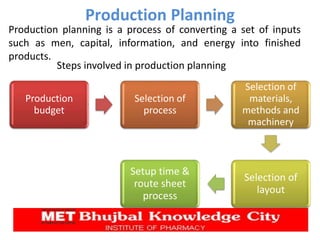
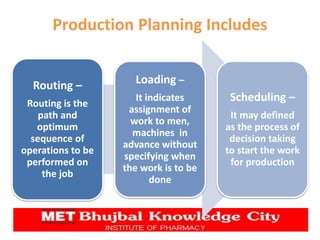
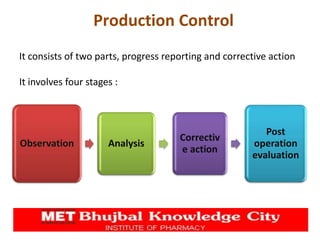
The document presents a comprehensive overview of pharmaceutical plant layout, detailing essential features, principles, types, and the factors affecting layouts for efficient production. It emphasizes the importance of planning for hygiene, minimizing cross-contamination, and ensuring optimal use of space and equipment to improve production quality and reduce costs. The document also highlights the processes of production planning, risk management, and production control to enhance operational efficiency.