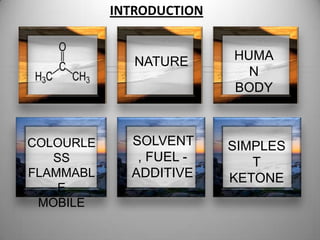
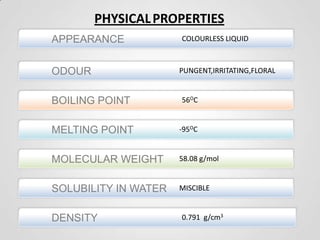
1. Acetone is a colorless, flammable, and mobile liquid that is the simplest ketone. It is used as a solvent and fuel additive.
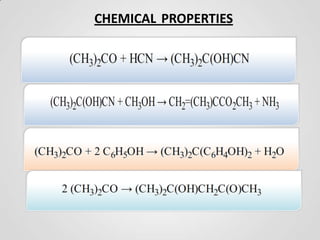
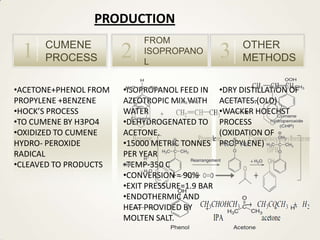
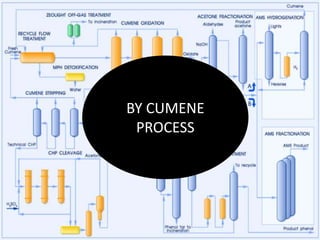
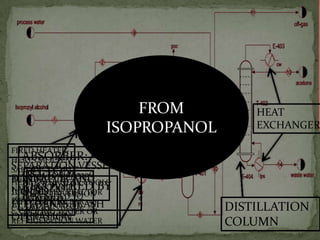
2. Acetone is produced commercially through two main processes - the cumene process and from isopropanol. The cumene process involves oxidizing cumene to form acetone and phenol. Production from isopropanol involves dehydrogenating isopropanol at high temperatures.
3. Acetone has a wide variety of applications including as a solvent for plastics, synthetic fibers, paints, adhesives, and varnishes. It is also used in cosmetics, laboratories, and as a miscellaneous solvent.