1) Before 1860, there was no standard system for determining atomic masses or numbers, leading to inconsistencies in proposed chemical compositions.
2) In 1860, Italian chemist Stanislao Cannizzaro presented a convincing method for accurately measuring atomic masses, which was agreed upon by chemists.
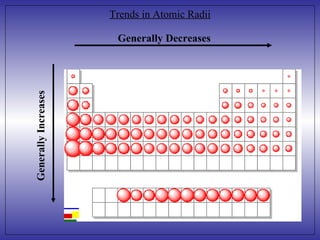
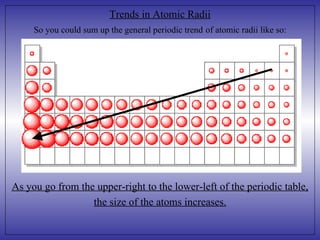
3) As you move from the upper-right to the lower-left of the periodic table, the size of atoms generally increases due to additional electrons being added as atomic number increases down groups and nuclear charge pulling electrons closer as atomic number increases across periods.