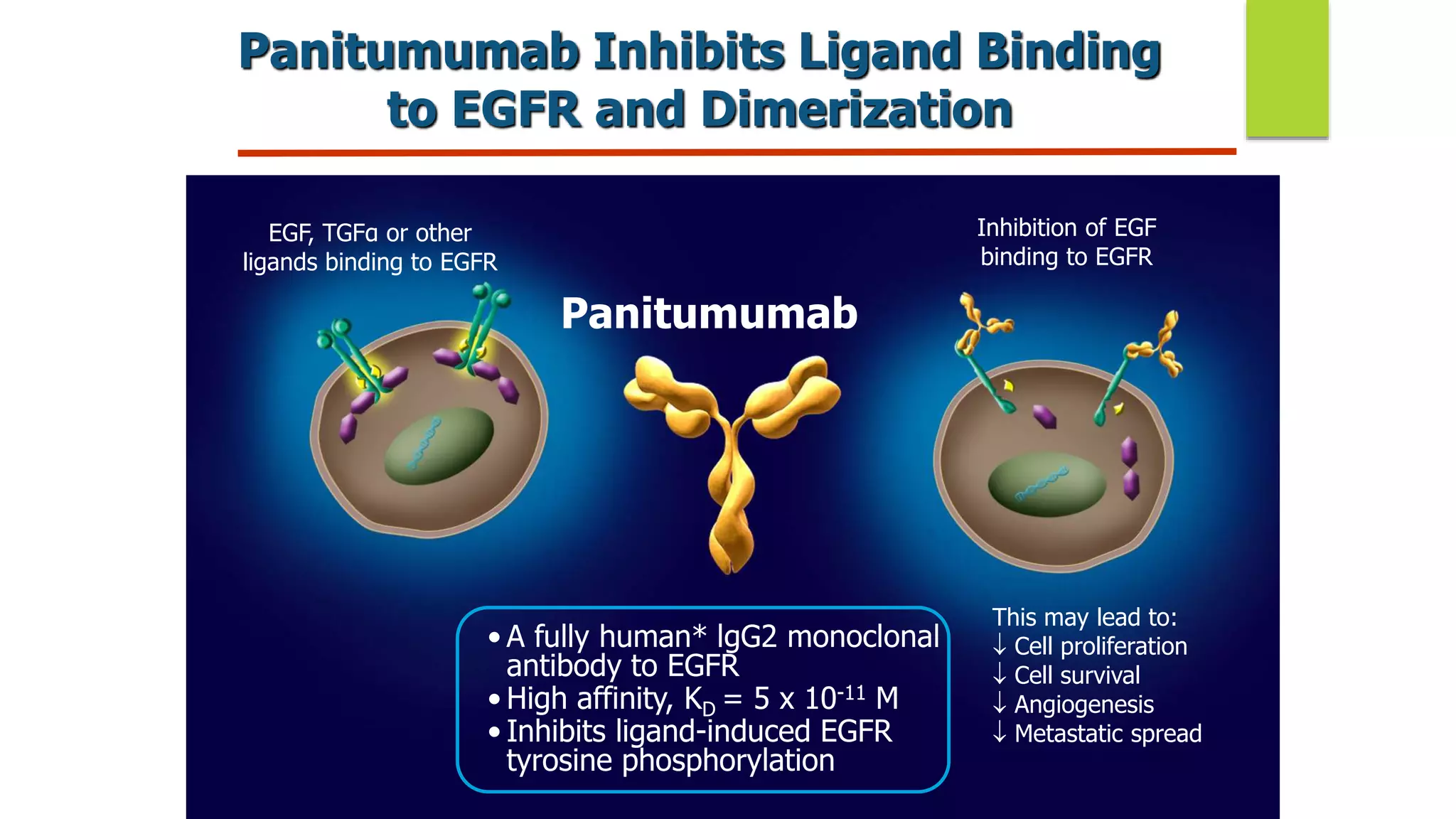
Panitumumab is a fully human monoclonal antibody that binds to the epidermal growth factor receptor (EGFR) with high affinity. It is approved as monotherapy or in combination with chemotherapy for wild-type KRAS metastatic colorectal cancer. Clinical trials have shown that panitumumab improves progression-free survival and overall survival when added to chemotherapy for first-line treatment of metastatic colorectal cancer patients with wild-type RAS tumors. Extended RAS testing beyond just KRAS exon 2 is important to identify patients most likely to benefit from panitumumab therapy. Common side effects include dermatological toxicities.