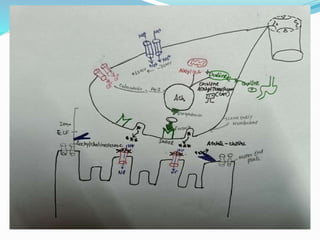
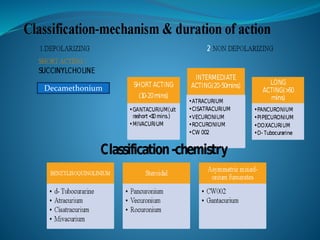
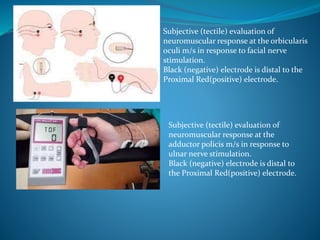
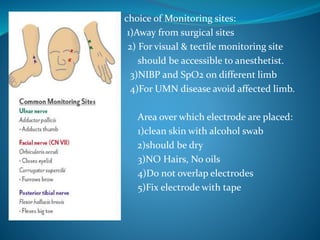
This document discusses pharmacology of neuromuscular blocking agents, their reversal, and neuromuscular monitoring. It begins by describing the neuromuscular junction and how it functions. It then discusses the mechanisms and effects of both depolarizing and non-depolarizing neuromuscular blocking agents. Specific agents like succinylcholine and atracurium are explained in detail. The document also covers neuromuscular monitoring techniques like train-of-four and how to interpret the responses. Reversal agents for neuromuscular blockade are also mentioned.




























![FACTORS THAT INCREASES THE POTENCY OF NON
DEPOLARISING NMBDs:
> Inhalational anaesthetics potentiates the neuromuscular blocking effect of non
depolarising NMBDs.
>Rank of order of potentiation : Desflurane > Sevoflurane, Isoflurane > Halothane
> Nitrous oxide – Barbiturates – Opoids – or Propofol anasthesia.
>Some antibiotics: Aminoglycosides, Lincomycin , Clindamycin, Polymixin.
>Hypothermia and Magnesium sulfate
>Combining two non depolarising NMBDs of chemically related drugs have
ADDITIVE effect [eg atracurium and mivacurium]
>Combining two structurally disimilar drugs have SYNERGISTIC effect [eg
rocuroniun and mivacurium]](https://image.slidesharecdn.com/nmjpresentation-240306161236-7e07656b/85/Neuromuscular-blocking-agents-Presentation-pptx-36-320.jpg)


![ADVERSE EFFECTS OF NEUROMUSCULAR BLOCKERS
A) AUTONOMIC EFFECTS: Tachycardia
Hypotension
Dysrythmias – pancuronium + halothane
Bradycardia – when combined with opiods, can even lead to asystole
–Tubocurarine administration is associated with marked ganglion
blockade resulting in hypotension
-Pancuronium has a direct vagolytic effect leading to tachycardia
B) HISTAMINE RELEASE:
Erythma of face, neck, and upper part of torso may develop, as well as
hypotenion and Reflex Tachycardia.
Can cause bronchospasm in patients with hyperactive airway disease.[
Rapacurium has highest incidence – withdrawn]
Seen with d-Tubocurarine, atracurium and mivacurium Histamine
release are decreased by slowing the injection rate.
TREATEMENT: Anti histaminics](https://image.slidesharecdn.com/nmjpresentation-240306161236-7e07656b/85/Neuromuscular-blocking-agents-Presentation-pptx-39-320.jpg)











