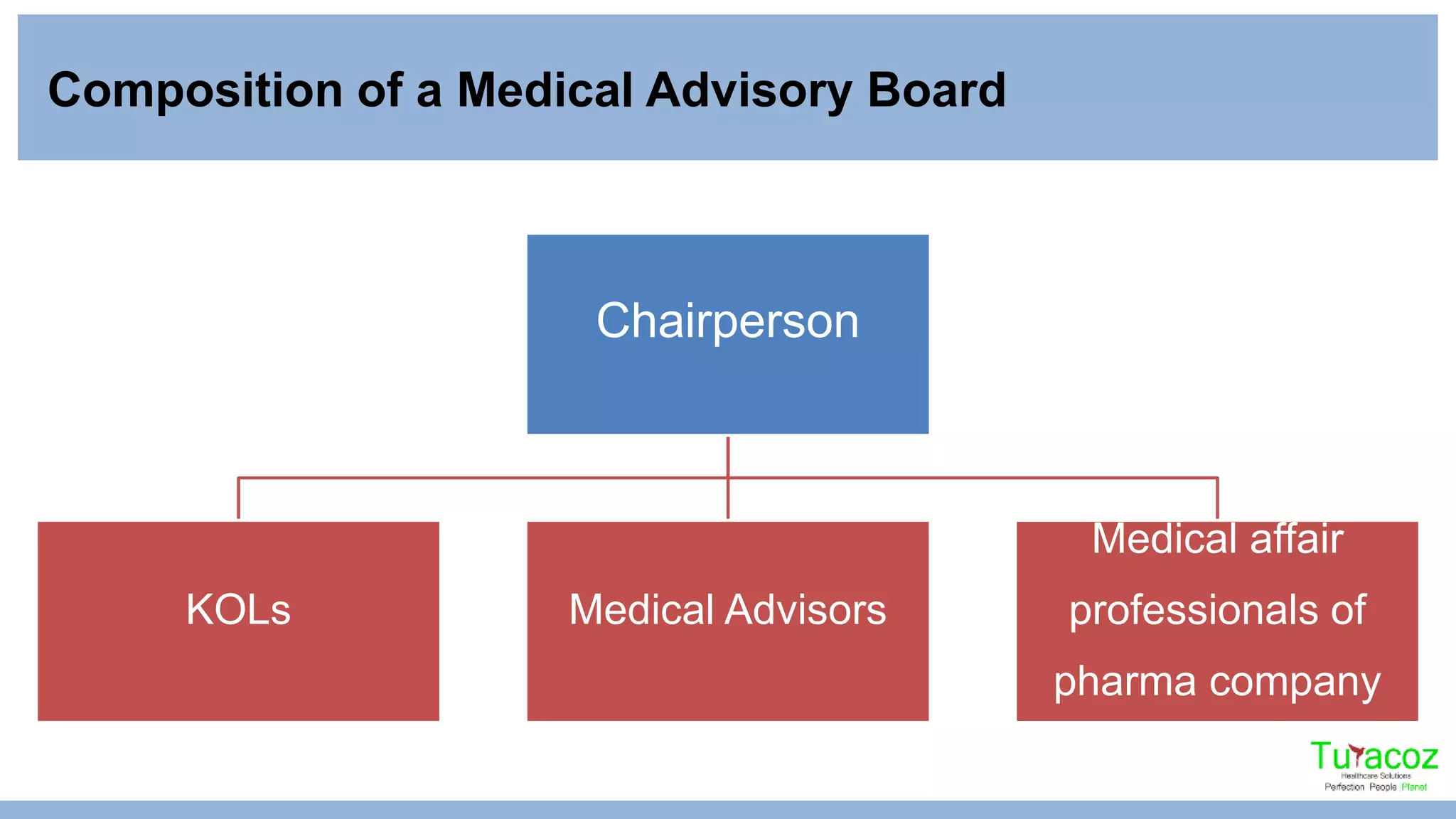
The document outlines the purpose and operations of medical advisory board meetings, highlighting their role in gathering insights from key opinion leaders (KOLs) regarding products or disease states. It details the responsibilities of medical writers in the process, including engaging KOLs, preparing reports and presentations, and documenting meeting minutes. It emphasizes the importance of thorough preparation and attentive participation to ensure effective knowledge sharing and actionable outcomes.