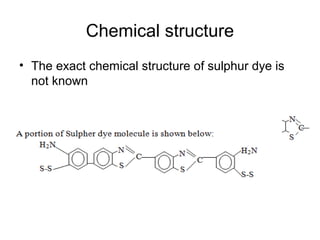
Sulphur dyes contain disulfide linkages and are used to produce black, brown, and other dark shades on cellulosic materials like cotton. They are water-insoluble so must be converted to a water-soluble leuco form using a reducing agent before application. After dyeing, an oxidizing agent turns the dye back to its insoluble form within the fiber. Issues like poor fastness, bronzing, and material tendering can occur but have corrective actions like ensuring complete dissolution and reaction of the dye and proper washing after dyeing.