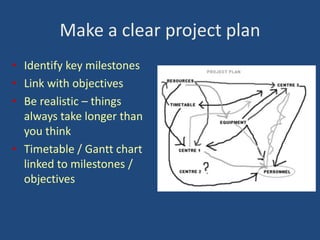
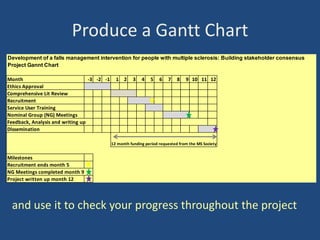
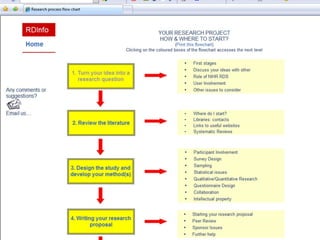
The document outlines the challenges and lessons learned by novice researchers conducting a study on fatigue management for multiple sclerosis. It highlights the importance of collaboration, securing funding, and planning in research, alongside the emotional ups and downs faced during the process. Key advice includes seeking support early, involving stakeholders, and being realistic about timelines and participant recruitment.