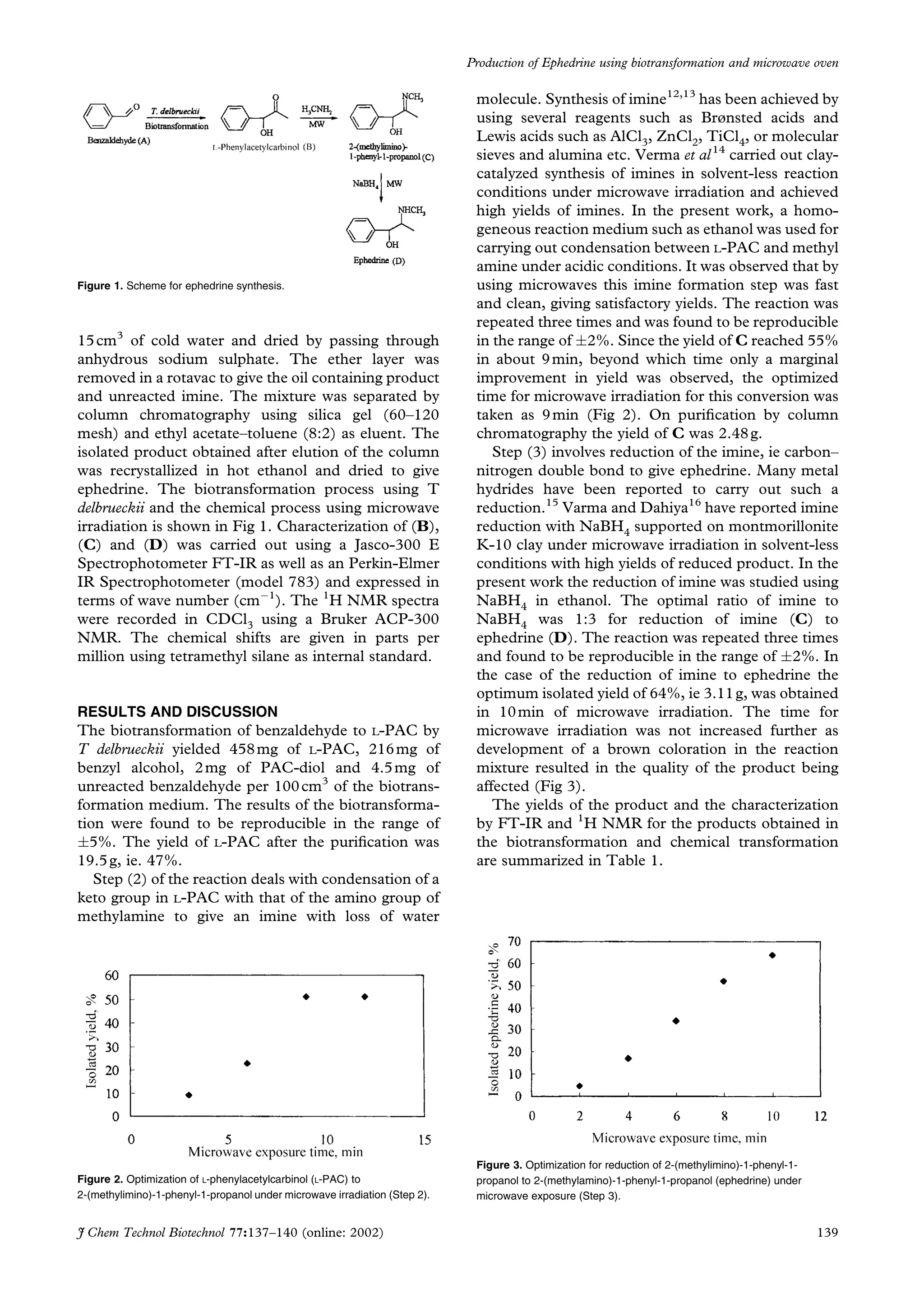
1) Torulaspora delbrueckii was used to biotransform 30g of benzaldehyde into 22.9g of L-phenylacetylcarbinol (L-PAC) in a 5L stirred tank reactor.
2) L-PAC was then converted to ephedrine in two microwave-assisted steps: first L-PAC was transformed to 2-(methylimino)-1-phenyl-1-propanol, then this was reduced to ephedrine.
3) The identities of the products were confirmed using 1H NMR and FT-IR analysis, demonstrating a facile synthesis of ephedrine from benzaldehyde using biocatalysis and microwave-assisted chemistry.