JBEI Research Highlights - January 2022
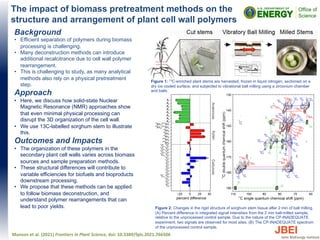
- 1. The impact of biomass pretreatment methods on the structure and arrangement of plant cell wall polymers Background • Efficient separation of polymers during biomass processing is challenging. • Many deconstruction methods can introduce additional recalcitrance due to cell wall polymer rearrangement. • This is challenging to study, as many analytical methods also rely on a physical pretreatment step. Approach • Here, we discuss how solid-state Nuclear Magnetic Resonance (NMR) approaches show that even minimal physical processing can disrupt the 3D organization of the cell wall. • We use 13C-labelled sorghum stem to illustrate this. Outcomes and Impacts • The organization of these polymers in the secondary plant cell walls varies across biomass sources and sample preparation methods. • These structural differences will contribute to variable efficiencies for biofuels and bioproducts downstream processing. • We propose that these methods can be applied to follow biomass deconstruction, and understand polymer rearrangements that can lead to poor yields. Munson et al. (2021) Frontiers in Plant Science, doi: 10.3389/fpls.2021.766506 Figure 1: 13C-enriched plant stems are harvested, frozen in liquid nitrogen, sectioned on a dry ice cooled surface, and subjected to vibrational ball milling using a zirconium chamber and balls. Figure 2: Changes in the rigid structure of sorghum stem tissue after 2 min of ball milling. (A) Percent difference in integrated signal intensities from the 2 min ball-milled sample, relative to the unprocessed control sample. Due to the nature of the CP-INADEQUATE experiment, two signals are observed for most sites. (B) The CP-INADEQUATE spectrum of the unprocessed control sample.
- 2. Background • Generating value from lignin through depolymerization and biological conversion is limited by lack of cost-effective depolymerization methods, toxicity within the breakdown products, and low bioconversion of the breakdown products. • High yield depolymerization of natural lignins requires cleaving carbon-carbon bonds in addition to ether bonds. Approach • To address that need, we report that a chelator-mediated Fenton reaction can efficiently cleave C-C bonds in sulfonated polymers at or near room temperature Outcomes and Impacts • This method was used to depolymerize lignosulfonate from Mw = 28,000 g/mol to Mw = 800 g/mol. The breakdown products were characterized by SEC, FTIR and NMR and evaluated for bioavailability. • A panel of nine organisms were tested for the ability to grow on the breakdown products. Growth at a low level was observed for several monocultures on the depolymerized lignosulfonate (LS) in absence of glucose. • Much stronger growth was observed in the presence of 0.2% glucose and for one organism we demonstrate doubling of melanin production in the presence of depolymerized LS. • The results suggest that this chelator-mediated Fenton method is a promising new approach for biological conversion of lignin into higher value chemicals or intermediates. Martinez et al. (2022) Green Chemistry, DOI: 10.1039/d1gc03854k Figure 1. Gel permeation chromatograms before and after treatment of lignosulfonate with chelated Fe(II) and H2O2. Depolymerization of lignin for biological conversion through sulfonaton and a chelator-mediated Fenton reaction Figure 2. Growth of microbes on depolymerized Lignosulfonate in the presence and absence of glucose Figure 5. Molecular weight distribution for [LS] = 5 mg/ml before and after reaction with [FeCl3] = [DHB] = 0.5 mM and [H2O2] = 0.5% at RT measured at a) 210 nm and b) 270 nm. a) b) Figure 10. Growth of monocultures in the LS breakdown stream or base medium in the presence or absence of glucose. Bars represent the average of three replicates and the error bars indicate the standard deviation.
- 3. Assembly of Artificial Metalloenzymes in Escherichia coli Nissle 1917 for Selectively Catalyzing Reactions Background • Artificial metalloenzymes (ArMs) are increasingly important in catalyzing abiological reactions with many examples in recent years, • Methods for assembling and screening ArMs are still laborious and time-consuming. • Assembly of ArMs in cells and screening them with the whole cell will significantly simplify and facilitate the creation and engineering processes. Approach • We used Escherichia coli Nissle 1917 (EcN) to create ArMs in vivo by expressing apo-P450 and importing the target artificial cofactor into cytoplasm, via a native transporter. • This strain was able to assemble the ArMs in vivo and selectively catalyze the reactions and provided the system to screen the directed evolution mutants of the ArMs. Outcomes and Impacts • This study developed an approach using a non-pathogenic E. coli strain to assemble ArMs in vivo, and significantly simply the process for creation of new ArMs and their improvement. • Catalyzing reactions and conducting directed evolution with this new microbial platform was tested and proved to be an efficient method. • A transporter able to import the target artificial cofactor was identified and can be used for further applications. Liu et al, J. Am. Chem. Soc., 2022, 144, 883-890 (A) Schematic diagram for the assembly of ArM in the EcN cells. (B) EcN cells harboring ArMs catalyzing site- selective C-H activation. (C) Identification of the cofactor transporter. A B 3 para-4 meta-4 + EcN harboring Ir-CYP119 N2 CO2Et O 30 oC, 4 h O CO2Et R O R R CO2Et + 3a R = Br CYP119-PL1 para:meta = 3.1:1 TON 2302±27 CYP119-ML1 para:meta = 1:3.2 TON 3241±54 3b R = Cl CYP119-PL1 para:meta = 2.8:1 TON 3407±234 CYP119-ML1 para:meta = 1:3.4 TON 4643±103 3c R = OMe 3d R = tBu 3a R = Br 3b R = Cl CYP119-PL1 para:meta = 4.9:1 TON 1634±36 CYP119-PL2 para:meta = 4.8:1 TON 938±140 C 1 2 + BL21(DE3) harboring Ir-CYP119 N2 CO2Et O 30 oC, 4 h O CO2Et
- 5. Heinz resistant tomato cultivars exhibit a lignin- based resistance to field dodder (Cuscuta campestris) parasitism Background • Cuscuta species (dodders) are agriculturally destructive, parasitic angiosperms. These parasitic plants use haustoria as physiological bridges to extract nutrients and water from hosts. • While some wild tomato relatives are resistant, cultivated tomatoes are generally susceptible to C. campestris infestations. However, some specific Heinz tomato (Solanum lycopersicum) hybrid cultivars exhibit resistance to dodders in the field, but their defense mechanism was previously unknown. Approach • We discovered that the stem cortex in these resistant lines responds with local lignification upon C. campestris attachment, preventing parasite entry into the host. LIF1 (Lignin Induction Factor 1, an AP2-like transcription factor), SlMYB55, and CuRLR1 (Cuscuta R-gene for Lignin-based Resistance 1, a CC-NBS-LRR) are identified as factors that confer host resistance by regulating lignification. Outcomes and Impacts • This work demonstrates how plants can utilize lignification to prevent parasitism by field dodder. • This discovery provides a foundation for investigating multilayer resistance against Cuscuta species and has potential for application in other essential crops attacked by parasitic plants. Jhu et al. (2022) Plant Physiology, doi: 10.1093/plphys/kiac024