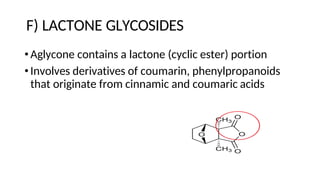
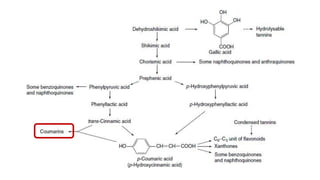
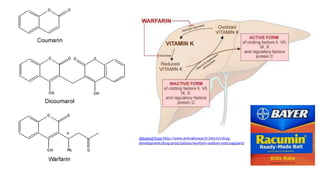
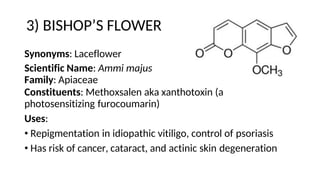
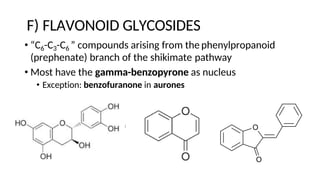
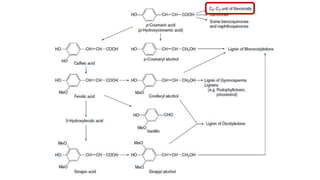
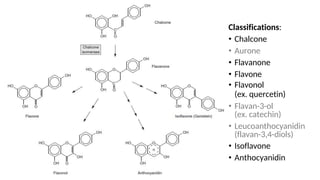
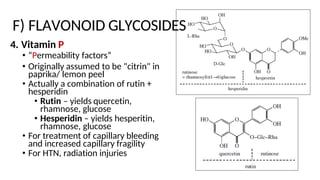
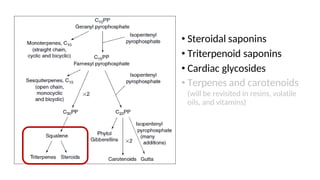
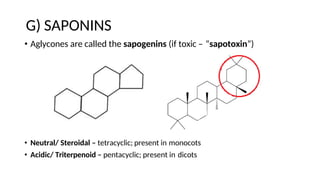
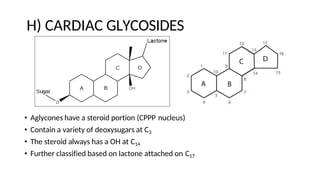
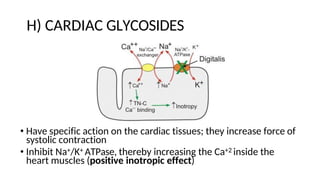
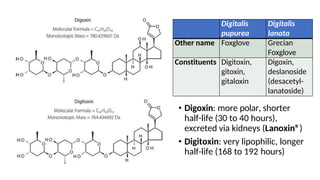
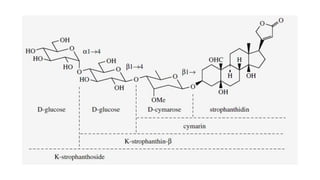
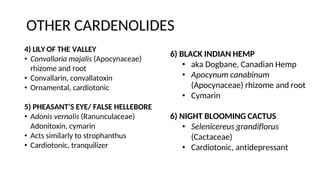
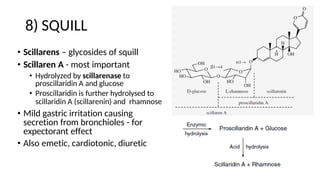
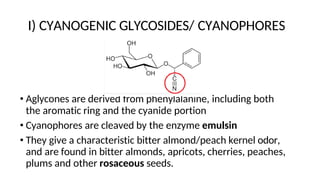
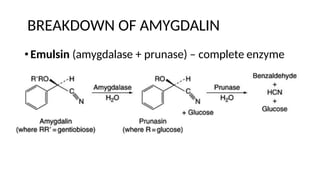
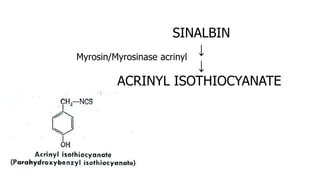
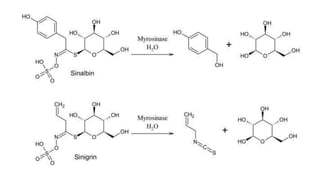
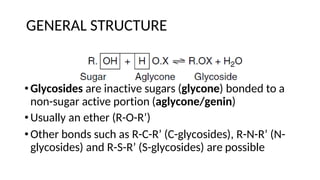
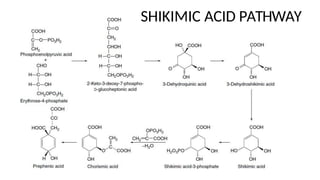
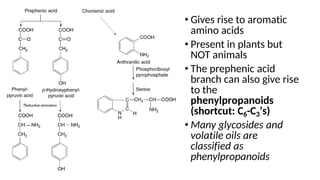
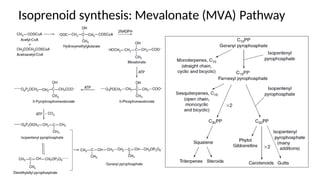
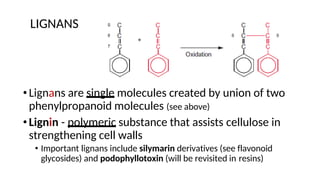
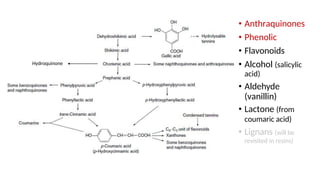
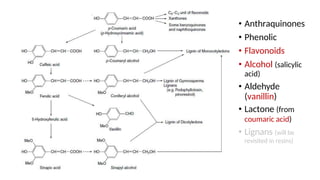
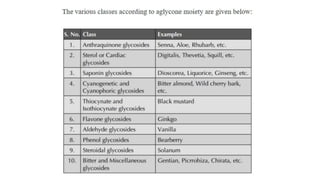
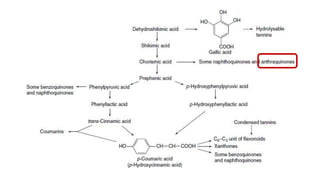
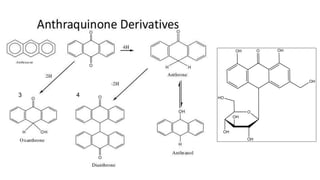
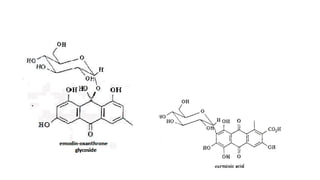
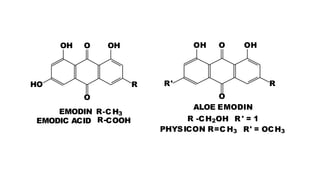
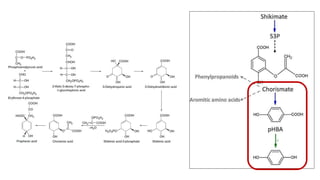
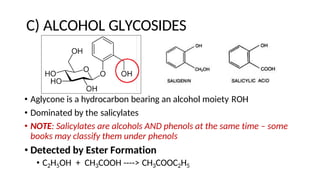
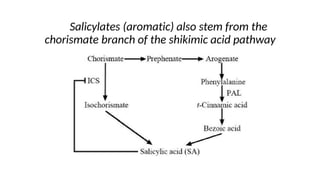
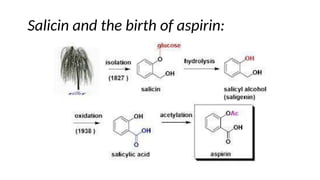
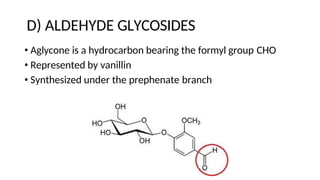
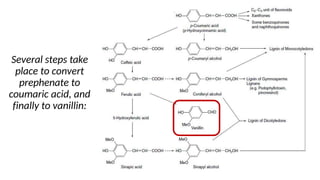
The document discusses various types of glycosides including their structures, biosynthesis pathways, and examples. It covers glycosides related to the shikimic acid pathway including anthraquinones, phenols, alcohols, aldehydes, and lactones. Specific examples described include cascara sagrada, senna, rhubarb, aloe, and vanilla. Glycosides related to the mevalonic acid pathway include saponins and cardiac glycosides. The document provides details on qualitative chemical tests used to identify different classes of glycoside aglycones.


















![7) ST. JOHN’S WORT (SJW)
Scientific Name: Hypericum perforatum
Family: Hypericaceae
Constituents: Hyperforin, hypericin
• Hyperforin is a prenylated ring –acts as reuptake inhibitor ->
increase neurotransmitters
• Was once dubbed as “Nature’s Prozac”
• [What is Prozac?]
• Claims to treat depression
• SJW is a well-known enzyme inducer](https://image.slidesharecdn.com/4glycosidesweek45-221219041834-d7874d4a/85/Glycosides-31-320.jpg)





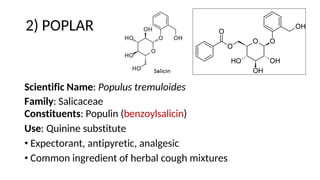

![1) VANILLA
• Unripe fruit of Vanilla planifolia (Mexican/ Bourbon
vanilla) or Vanilla tahitensis (Tahitian) [Orchidaceae]
• Contains glucovanillin (avenein)
• Vanilla must undergo sweating to release odor (hydrolysis
of glucovanillin to yield vanillin)
• Vanillin can be artificially made using eugenol (clove) or more
commonly today, guiacol (Pinaceae)
• Vastly used in food, perfumery, and cosmetics
• Vanilla is the SECOND most expensive spice in the world!
• Mexican/Vera cruz – best variety
• Bourbon – coumarinlike odor
• Tahiti – reddish brown, unpleasant odor](https://image.slidesharecdn.com/4glycosidesweek45-221219041834-d7874d4a/85/Glycosides-45-320.jpg)