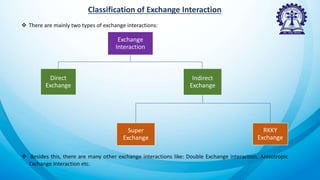
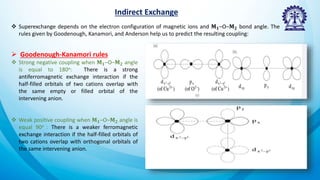
The document discusses exchange interactions, their origins, classifications, and effects on magnetic properties in quantum mechanics. It elaborates on direct and indirect exchange, outlining the mechanisms of super exchange, RKKY exchange, double exchange, and anisotropic exchange interactions. These interactions primarily influence behavior in magnetic materials, emphasizing the significance of orbital overlaps and the role of different bond angles in determining coupling strengths.



![Origin of Exchange Interaction
f
Consider a simple model with just two electrons (𝐬𝟏= 𝐬𝟐=
𝟏
𝟐
).
So, total spin, S =
1
2
−
1
2
, (
1
2
+
1
2
) = 0 (singlet),1 (triplet)
Singlet State :
S = 0. So, 𝐦𝐬 = 0 → only one state
Total wave function, 𝚿𝐬 = 𝛗𝐬 𝛘𝐬
𝛗𝐬 →symmetric, 𝛘𝐬 →antisymmetric (since 𝚿𝐬 →antisymmetric).
Here, 𝛗𝐬 =
𝟏
𝟐
[ 𝝋𝒂(𝑟𝑎) 𝝋𝒃(𝑟𝑏) + 𝝋𝒂(𝑟𝑏) 𝝋𝒃(𝑟𝑎) ] and
𝛘𝐬 =
𝟏
𝟐
[ ↑ ↓ - ↓ ↑ ]
Energy: 𝐄𝐒 = 𝚿𝐬
∗
𝐇 𝚿𝐬 d𝐫𝟏 d𝐫𝟐 and 𝐬𝟏.𝐬𝟐 = -
𝟑
𝟒
Triplet States :
S = 1. So, 𝐦𝐬 = -1,0,+1 → total three states
Total wave function, 𝚿𝐓 = 𝛗𝐓 𝛘𝐓
𝛗𝐓 →antisymmetric, 𝛘𝐓 →symmetric (since 𝚿𝐓 →antisymmetric).
Here, 𝛗𝐓 =
𝟏
𝟐
[ 𝝋𝒂(𝑟𝑎) 𝝋𝒃(𝑟𝑏) - 𝝋𝒂(𝑟𝑏) 𝝋𝒃(𝑟𝑎) ] and
𝛘𝐓 = ↑ ↑ , ↓ ↓ ,
𝟏
𝟐
[ ↑ ↓ + ↓ ↑ ]
Energy: 𝐄𝐓 = 𝚿𝐓
∗
𝐇 𝚿𝐓 d𝐫𝟏 d𝐫𝟐 and 𝐬𝟏.𝐬𝟐 =
𝟏
𝟒](https://image.slidesharecdn.com/exchangeinteractionandtheirconsequences-221112123829-7f254a29/85/Exchange-Interaction-and-their-Consequences-pptx-4-320.jpg)