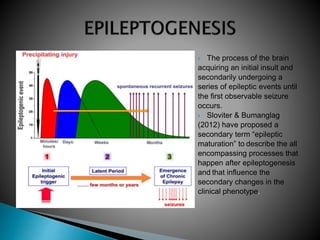
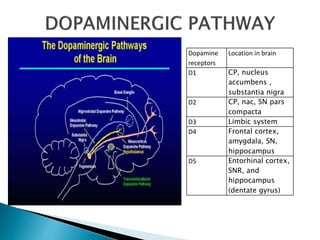
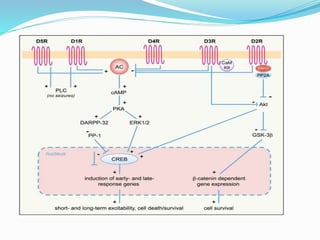
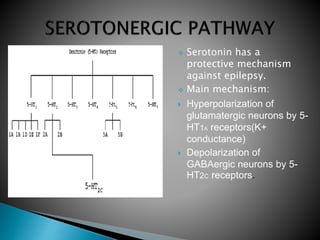
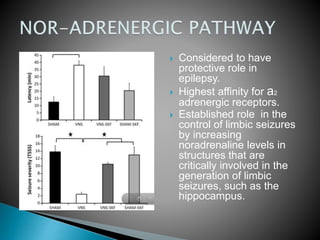
1) Epilepsy is defined as two or more unprovoked seizures occurring more than 24 hours apart or one unprovoked seizure with a high risk of further seizures in the next 10 years.
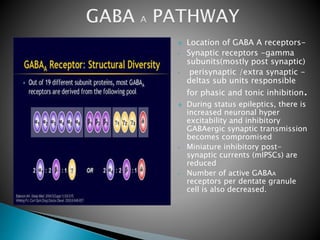
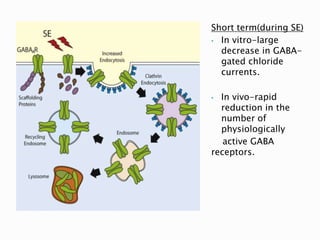
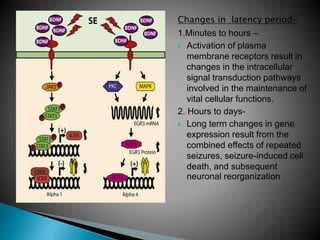
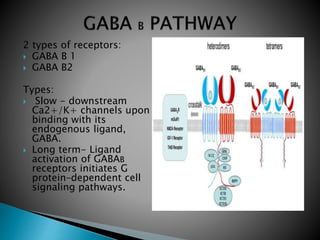
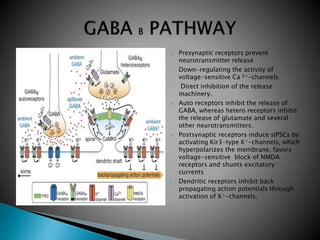
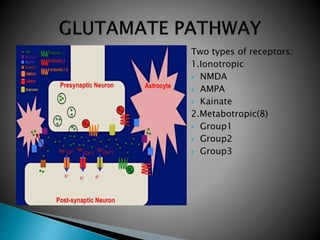
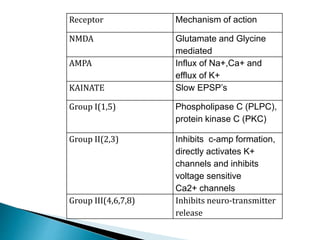
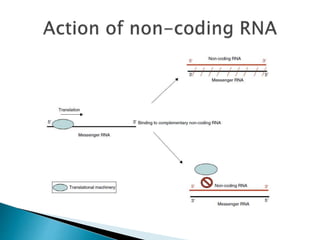
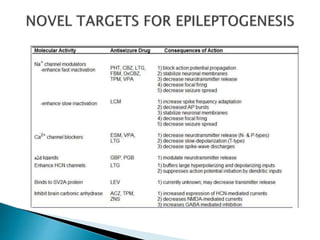
2) GABA, glutamate, and other neurotransmitters play a role in epileptogenesis. During seizures, GABAergic inhibition is compromised while glutamatergic excitation increases.
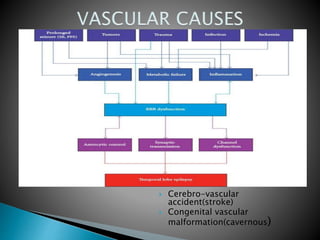
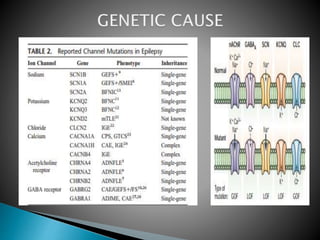
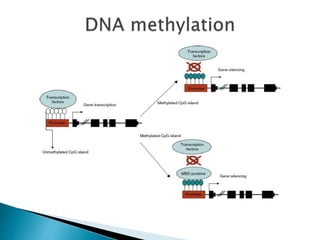
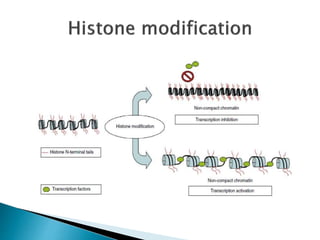
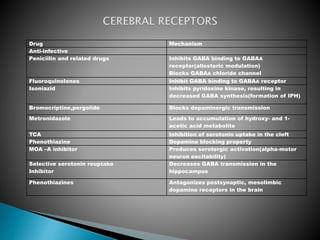
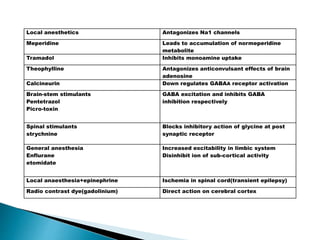
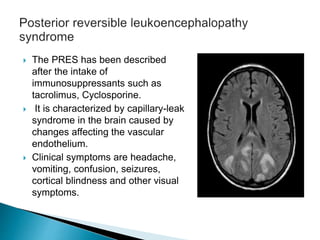
3) Many factors can cause seizures including vascular, metabolic, infectious, autoimmune, genetic, and drug-induced causes by altering excitatory and inhibitory neurotransmission in the brain.