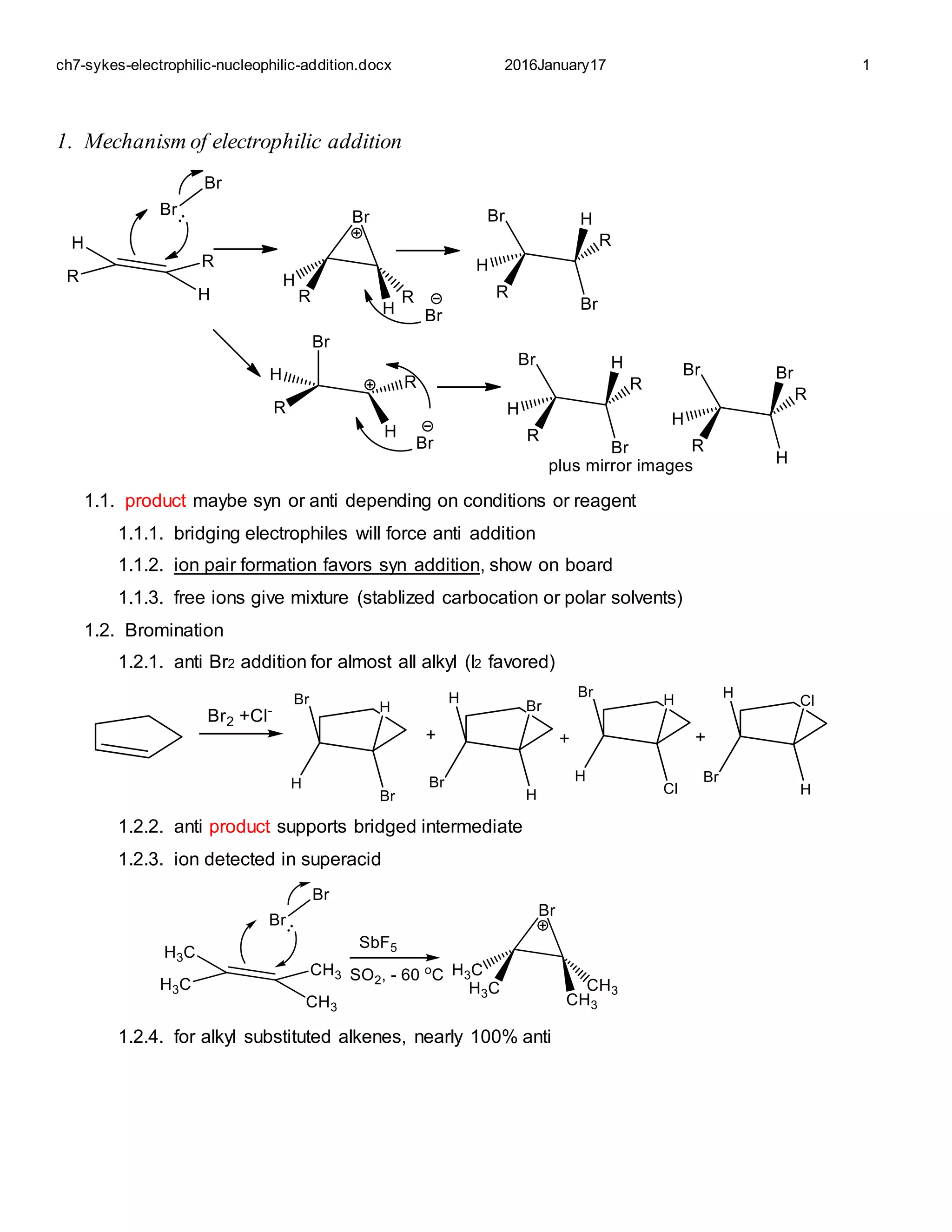
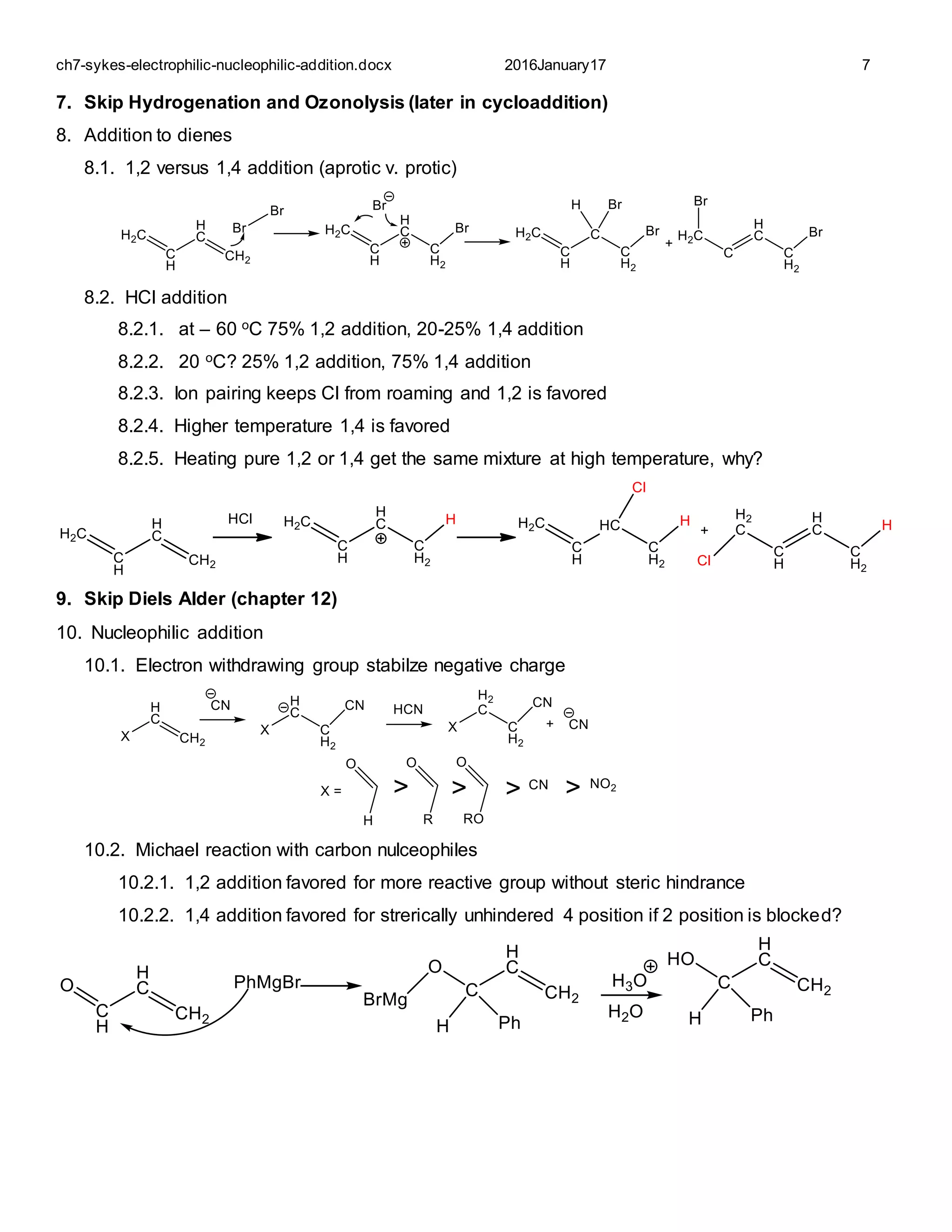
This document summarizes various mechanisms of electrophilic and nucleophilic addition to alkenes. It discusses factors that determine the stereochemistry of addition, including the use of bridging electrophiles or ion pairing. It also covers addition of halides, protons, water, hydroboration-oxidation, carbon cations, peroxy acids, and nucleophiles. The key factors that determine the stereochemistry and regioselectivity of these addition reactions are discussed.