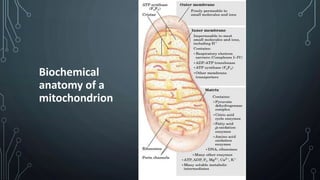
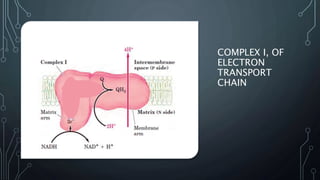
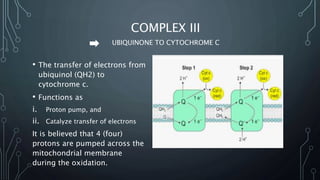
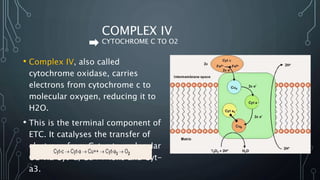
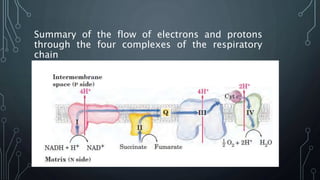
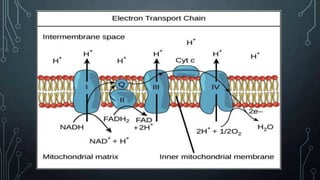
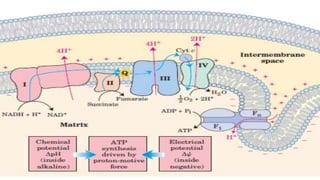
The document discusses the electron transport chain and oxidative phosphorylation, detailing how they re-oxidize NADH and FADH2 to trap energy as ATP. It explains the structure and function of the four complexes of the electron transport chain in mitochondria and describes the proton motive force that drives ATP synthesis. The document also highlights the potential use of the proton gradient for heat generation in certain cells, such as brown fat cells in hibernating mammals.