Diffusion tensor imaging (DTI) allows the measurement of water diffusion in tissues to probe microstructure beyond image resolution. DTI characterizes diffusion anisotropy, in which diffusion varies with direction. In white matter, diffusion is fastest along axonal fibers and slower perpendicular, revealing fiber orientation. DTI exploits this to map white matter tracks in the brain. The article reviews DTI concepts and applications like fiber tracking, which combined with functional MRI may provide insights into brain connectivity.

![Diffusion Tensor Imaging 535
nates. Diffusion anisotropy in white matter originates
from its specific organization in bundles of more or less
myelinated axonal fibers running in parallel, although
the exact mechanism is still not completely understood
(see below): diffusion in the direction of the fibers is
faster than in the perpendicular direction. It quickly
appeared that this feature could be exploited to map
out the orientation in space of the white matter tracks
in the brain using a color scale, assuming that the
direction of the fastest diffusion would indicate the
overall orientation of the fibers (15).
This pioneering work on diffusion anisotropy really
took off with the introduction of the more rigorous for-
malism of the diffusion tensor by Basser et al. (16 –18).
With diffusion tensor imaging (DTI), diffusion anisot-
ropy effects in diffusion MRI data could be fully ex-
tracted, characterized, and exploited, providing even
more exquisite details of tissue microstructure. Many
studies have been published dealing with the optimiza-
tion of the MRI sequences necessary to gain access to
the diffusion tensor, the processing and display of DTI
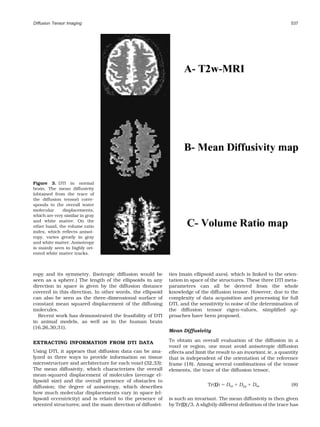
Figure 1. The diffusion tensor, axial slices. Diffusivity along x,
data, and, of course, potential applications. The most
y, and z axes, shown in Dxx (a), Dyy (d), and Dzz (f) images,
advanced application is certainly that of fiber tracking
respectively, is clearly different in white matter, especially in
in the brain, which, in combination with functional the corpus callosum. Nondiagonal images [Dxy (b), Dxz (c), and
MRI, might open a window onto the important issue of Dyz (e)] are not noise images because the x, y, z reference frame
connectivity. of the MRI scanner does not coincide with the diffusion refer-
The aim of this article is to review the concepts behind ence frame of tissues in most voxels.
DTI and to present potential applications.
DIFFUSION TENSOR MRI: DATA ACQUISITION where bii are the elements of the b matrix (which now
AND PROCESSING replaces the b factor) expressed in the coordinates of
The Diffusion Tensor this reference frame.
In practice, unfortunately, measurements are made
With plain diffusion MRI, diffusion is fully described
in the reference frame [ x, y, z] of the MRI scanner
using a single (scalar) parameter, the diffusion coeffi-
gradients, which usually does not coincide with the
cient, D. The effect of diffusion on the MRI signal (most
diffusion frame of the tissue. Therefore, one must also
often a spin-echo signal) is an attenuation, A, which
consider the coupling of the nondiagonal elements, bij,
depends on D and on the “b factor,” which characterizes
of the b matrix with the nondiagonal terms, Dij, (i j),
the the gradient pulses (timing, amplitude, shape) used
of the diffusion tensor (now expressed in the scanner
in the MRI sequence (8):
frame), which reflect correlation between molecular dis-
placements in perpendicular directions (17):
A exp bD . (1)
However, in the presence of anisotropy, diffusion can A exp bijDij (4)
no longer be characterized by a single scalar coefficient, i x,y,z j x,y,z
but requires a tensor, D, which fully describes molecu-
lar mobility along each direction and correlation be- or
tween these directions (4) (Fig. 1):
A exp bxxDxx byyDyy bzzDzz
Dxx Dxy Dxz
D Dyx Dyy Dyz . (2) 2bxyDxy 2bxzDxz 2byzDyz . (5)
Dzx Dzy Dzz
Hence, it is important to note that by using diffusion-
This tensor is symmetric (Dij Dji, with i, j x, y, z). encoding gradient pulses along one direction only, say
In a reference frame [ x , y , z ] that coincides with the x, signal attenuation not only depends on the diffusion
principal or self directions of diffusivity, the off-diagonal effects along this direction but may also include contri-
terms do not exist, and the tensor is reduced only to its bution from other directions, say y and z.
diagonal terms, Dx x , Dy y , Dz z , which represent molec- Calculation of b may quickly become complicated
ular mobility along axes x , y , and z , respectively. The when many gradient pulses are used (20,21), but the
echo attenuation then becomes: full determination of the diffusion tensor is necessary if
one wants to assess properly and fully all anisotropic
A exp bx x Dx x by y Dy y bz z Dz z (3) diffusion effects.](https://image.slidesharecdn.com/dtibasics-110224131255-phpapp02/85/Dti-basics-2-320.jpg)
![536 Le Bihan et al.
DTI Data Acquisition
To determine the diffusion tensor fully, one must first
collect diffusion-weighted images along several gradi-
ent directions, using diffusion-sensitized MRI pulse se-
quences (22) such as echoplanar imaging (EPI). As the
diffusion tensor is symmetric, measurements along
only six directions are mandatory (instead of nine),
along with an image acquired without diffusion weight-
ing (b 0). A typical set of gradient combinations that
preserves uniform space sampling and similar b values
along each direction is as follows (coefficients for gradi-
ent pulses along the (x, y, z) axes, normalized to a given
amplitude, G):
1/ 2, 0, 1/ 2 ; 1/ 2, 0, 1/ 2 ;
0, 1/ 2, 1/ 2 ; 0, 1/ 2, 1/ 2 ;
1/ 2, 1/ 2, 0 ; 1/ 2, 1/ 2, 0 .
This minimal set of images may be repeated for averag-
ing, as the signal-to-noise ratio (SNR) may be low
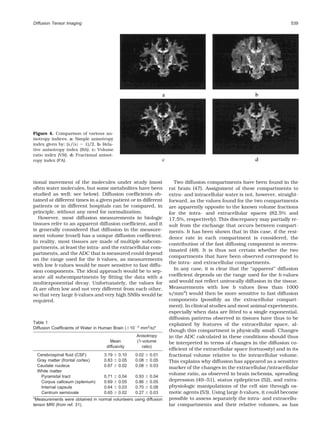
(17,18). Note that it is not necessary (and not efficient in Figure 2. Schemes for directional sampling. Although initial
terms of SNR) to obtain data with different b values for sampling schemes consisted of six (or even four) directions of
each direction. A simple calculation (22) shows that, in measurements, progress is made in scanning space more uni-
the case of scalar diffusion, one gets the best accuracy formly along many directions, especially for fiber orientation
on the diffusion coefficient, D, from two acquisitions mapping.
when using 2 b factors, b1 and b2, differing by about
1/D (23). In the brain, this translates to (b2 b1)
1000–1500 s/mm2. If more than two acquisitions are to
multiple linear regression, using Eq. [5]. From the dif-
be used (for averaging purposes), error propagation the-
fusion tensor components, one may calculate indices
ory (24) shows that it is better to accumulate n1 and n2
that reflect either the average diffusion or the degree of
measurements at each of the low and high b factors, b1
anisotropy in each voxel (see below). Another step is to
and b2, than to use a range of b factors. The accuracy
determine the main direction of diffusivities in each
dD/D is then obtained from the raw image SNR:
voxel and the diffusion values associated with these
1/2 directions. This is equivalent to determining the refer-
dD/D 1/n 1 exp 2D b1 b2 /n2
ence frame [ x , y , z ] where the off-diagonal terms of D
SNR D b1 b2 . (6) are null. This “diagonalization” of the diffusion tensor
provides eigen-vectors and eigen-values, , which cor-
In the anisotropic case, this paradigm would be still respond respectively to the main diffusion directions
useful, but one should bear in mind that D will change and associated diffusivities (17,18). (The eigen-diffu-
for each voxel according to the measurement direction sivities, , are not to be confused with the tortuosity
(25). factors.)
Acquisition performed with linear sets of b value As it is difficult to display tensor data with images
ranges remains, however, extremely useful to assess (multiple images would be necessary), the concept of
the sequence/hardware performance (stability, eddy diffusion ellipsoids has been proposed (17,18). An ellip-
currents, . . . ) (22) or to characterize the diffusion pro- soid is a three-dimensional representation of the diffu-
cess better, in particular in the presence of multiple sion distance covered in space by molecules in a given
diffusion compartments (see below). diffusion time, Td. These ellipsoids, which can be dis-
In the case of axial symmetry, only four directions are played for each voxel of the image, are easily calculated
necessary (tetrahedral encoding) (26), as suggested in from the eigen diffusivities, 1, 2, and 3 (correspond-
the spinal cord (27,28). The acquisition time and the ing to Dx x , Dy y , and Dz z ):
number of images to process are then reduced. In con-
trast, efforts are now being made to collect data along as x 2/ 2 Td y 2/ 2 Td z 2/ 2 Td 1 (7)
1 2 3
many directions in space as possible to avoid sampling
direction biases. This uniform space sampling para- where x , y , and z refer to the frame of the main
digm is particularly interesting for fiber tracking appli- diffusion direction of the tensor. These eigen diffusivi-
cations and provides a gain in SNR (25,29) (Fig. 2 and ties represent the unidimensional diffusion coefficients
see below). in the main directions of diffusivities of the medium.
Therefore, the main axis of the ellipsoid gives the main
DTI Data Processing diffusion direction in the voxel (coinciding with the di-
A second step is to estimate the Dij values from the set rection of the fibers), while the eccentricity of the ellip-
of diffusion/orientation-weighted images, generally by soid provides information about the degree of anisot-](https://image.slidesharecdn.com/dtibasics-110224131255-phpapp02/85/Dti-basics-3-320.jpg)
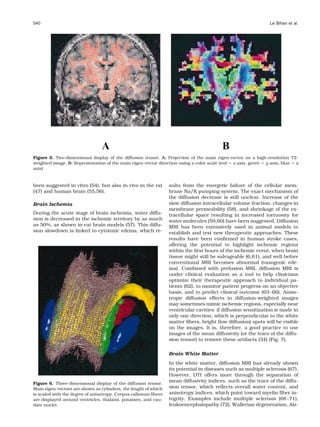
![538 Le Bihan et al.
proved useful in assessing the diffusion drop in brain RA, a normalized standard deviation, also represents
ischemia (34) (Fig. 3 and see below). the ratio of the anisotropic part of D to its isotropic part.
Unfortunately, the correct estimation of Tr(D) still FA measures the fraction of the “magnitude” of D that
requires the complete determination of the diffusion can be ascribed to anisotropic diffusion. FA and RA vary
tensor. Generally, one cannot simply add diffusion co- between 0 (isotropic diffusion) and 1( 2 for RA) (infinite
efficients obtained by separately acquiring data with anisotropy). As for VR, which represents the ratio of the
gradient pulses added along x, y, and z axes, as these ellipsoid volume to the volume of a sphere of radius ,
measured coefficients usually do not coincide with Dxx, its range is from 1 (isotropic diffusion) to 0, so that some
Dyy, and Dzz, respectively (see above). The reason is that authors prefer to use (1 VR) (39).
the diffusion attenuation that results, for instance, Once these indices have been defined, it is possible to
from inserting gradients on the x axis (see above) is not evaluate them directly from diffusion-weighted images,
simply A exp[ bxxDxx], unless diffusion is isotropic ie, without the need to calculate the diffusion tensor
(no nondiagonal terms) or the contribution of the gra- (40). For instance, A , which is very similar to RA, has
dient pulses on y and z axes is negligible. (Sequences been proposed as (41):
can be written that way.)
To avoid this problem and to simplify the approach, A Di D 2 2
D xy 2
D xz D2 / 6 D
yz
several groups have designed sequences based on mul- i x,y,z
tiple echoes or acquisitions with tetrahedral gradient (13)
configurations, so as to cancel nondiagonal term con-
tributions to the MRI signal directly (26,35–37). with
Diffusion Anisotropy Indices D D i /3 (14)
i x,y,z
Several scalar indices have been proposed to character-
ize diffusion anisotropy. Initially, simple indices calcu-
Also, images directly sensitive to anisotropy indices, or
lated from diffusion-weighted images (10) or ADCs ob-
anisotropically weighted images, can be obtained (42).
tained in perpendicular directions were used, such as
Finally, the concept of these intravoxel anisotropy indi-
ADCx/ADCy (15) and displayed using a color scale.
ces can be extended to a family of intervoxel or “lattice”
Other groups have devised indices mixing measure-
measures of diffusion anisotropy, which allows neigh-
ments along x, y, and z directions, such as max[ADCx,
boring voxels to be considered together, in a region of
ADCy, ADCz]/min[ADCx, ADCy, ADCz] or the standard
interest, without losing anisotropy effects resulting
deviation of ADCx, ADCy, and ADCz divided by their
from different fiber orientations across voxels (39). Clin-
mean value (34). Unfortunately, none of these indices
ically relevant images of anisotropy indices have been
are really quantitative, as they do not correspond to a
obtained in the human brain (31,43) (Figs. 3, 4; Ta-
single meaningful physical parameter and, more impor-
ble 1).
tantly, are clearly dependent on the choice of directions
made for the measurements. The degree of anisotropy
would then vary according to the respective orientation Fiber Orientation Mapping
of the gradient hardware and the tissue frames of ref- The last family of parameters that can be extracted from
erence and would generally be underestimated. Here the DTI concept relates to the mapping of the orienta-
again, invariant indices must be found to avoid such tion in space of tissue structure. The assumption is that
biases and provide an objective, intrinsic structural the direction of the fibers is colinear with the direction
information (38). of the eigen-vector associated with the largest eigen
Invariant indices are thus made of combinations of diffusivity. This approach opens a completely new way
the terms of the diagonalized diffusion tensor, ie, the to gain direct and in vivo information on the organiza-
eigen-values 1, 2, and 3. The most commonly used tion in space of oriented tissues, such as muscle, myo-
invariant indices are the relative anisotropy (RA), the cardium, and brain or spine white matter, which is of
fractional anisotropy (FA), and the volume ratio (VR) considerable interest, clinically and functionally. Direc-
indices, defined respectively as: tion orientation can be derived from DTI directly from
diffusion/orientation-weighted images or through the
RA 1
2
2
2
3
2
/ 3 calculation of the diffusion tensor. A first issue is to
display fiber orientation on a voxel-by-voxel basis. The
(9) use of color maps has first been suggested (15), followed
where by representation of ellipsoids (18,39), octahedra (44),
or vectors pointing in the fiber direction (45,46) (Figs.
5, 6).
1 2 3 /3. (10)
2 2 2 APPLICATIONS
FA 3 1 2 3 /
2 2 2
It is important to notice that diffusion imaging is a truly
2 1 2 3 . (11) quantitative method. The diffusion coefficient is a phys-
ical parameter that directly reflects the physical prop-
3
VR 1 2 3 / . (12) erties of the tissues, in terms of the random transla-](https://image.slidesharecdn.com/dtibasics-110224131255-phpapp02/85/Dti-basics-5-320.jpg)


![Diffusion Tensor Imaging 545
56. Clark CA, Le Bihan D. Water diffusion compartmentation and 80. Buchsbaum MS, Tang CY, Peled S, et al. MRI white matter diffu-
anisotropy at high b values in human brain. In: Proceedings of the sion anisotropy and PET metabolic rate in schizophrenia. Neuro-
SMRM, 2000. p 759. report 1998;9:425– 430.
57. Moseley ME, Kucharczyk J, Mintorovitch J, et al. Diffusion- 81. Foong J, Maier M, Clark CA, et al. Neuropathological abnormali-
weighted MR imaging of acute stroke: correlation with T2- ties of the corpus callosum in schizophrenia. J Neurol Neurosurg
weighted and magnetic susceptibility-enhanced MR imaging in Psychiatry 2000;68:242–244.
cats. AJNR 1990;11:423– 429. 82. Klingberg T, Hedehus M, Temple E, et al. Microstructure of tem-
58. Latour LL, Svoboda K, Mitra PP, Sotak CH. Time-dependent dif- poro-parietal white matter as a basis for reading ability: evidence
fusion of water in a biological model system. Proc Natl Acad Sci from diffusion tensor magnetic resonance imaging. Neuron 2000;
USA 1994;91:1229 –1233. 25:493–500.
59. Norris DG, Niendorf T, Leibfritz D. Healthy and infarcted brain 83. Le Bihan D, Douek P, Argyropoulou M, et al. Diffusion and perfu-
tissues studied at short diffusion times: the origins of apparent sion magnetic resonance imaging in brain tumors. Top Magn
restriction and the reduction in apparent diffusion coefficient. Reson Imaging 1993;5:25–31.
NMR Biomed 1994;7:304 –310. 84. Ikezaki K, Takahashi M, Koga H, et al. Apparent diffusion coeffi-
60. Pfeuffer J, Dreher W, Sykova E, Leibfritz D. Water signal attenu- cient (ADC) and magnetization transfer contrast (MTC) mapping of
ation in diffusion-weighted 1H NMR experiments during cerebral experimental brain tumor. Acta Neurochir Suppl (Wien) 1997;70:
ischemia: influence of intracellular restrictions, extracellular tor- 170 –172.
tuosity, and exchange. Magn Reson Imaging 1998;16:1023–1032. 85. Krabbe K, Gideon P, Wagn P, et al. MR diffusion imaging of human
61. Sorensen AG, Buonanno FS, Gonzalez RG, et al. Hyperacute intracranial tumours. Neuroradiology 1997;39:483– 489.
stroke: evaluation with combined multisection diffusion-weighted 86. Barzo P, Marmarou A, Fatouros P, Hayasaki K, Corwin F. Contri-
and hemodynamically weighted echo-planar MR imaging. Radiol- bution of vasogenic and cellular edema to traumatic brain swelling
ogy 1996;199:391– 401. measured by diffusion-weighted imaging. J Neurosurg 1997;87:
62. Warach S, Boska M, Welch KMA. Pitfalls and potential of clinical 900 –907.
diffusion-weighted MR imaging in acute stroke. Stroke 1997;28: 87. Schwartz RB, Mulkern RV, Gudbjartsson H, Jolesz F. Diffusion-
481– 482. weighted MR imaging in hypertensive encephalopathy: clues to
63. Lovblad KO, Baird AE, Schlaug G, et al. Ischemic lesion volumes
¨ pathogenesis. AJNR 1998;19:859 – 862.
in acute stroke by diffusion-weighted magnetic resonance imaging 88. Chang L, Ernst T. MR spectroscopy and diffusion-weighted MR
correlate with clinical outcome. Ann Neurol 1997;42:164 –170. imaging in focal brain lesions in AIDS. Neuroimaging Clin N Am
64. Gonzalez RG, Schaefer PW, Buonanno FS, et al. Diffusion- 1997;7:409 – 426.
weighted MR imaging: diagnostic accuracy in patients imaged 89. Schaefer PW, Buonanno FS, Gonzalez RG, Schwamm LH. Diffu-
within 6 hours of stroke symptom onset. Radiology 1999;210:
sion-weighted imaging discriminates between cytotoxic and vaso-
155–162.
genic edema in a patient with eclampsia. Stroke 1997;28:1082–
65. Warach S, Boska M, Welch KM. Pitfalls and potential of clinical
1085.
diffusion-weighted MR imaging in acute stroke [editorial; com-
90. Okada K, Wu LH, Kobayashi S. Diffusion-weighted MRI in severe
ment]. Stroke 1997;28:481– 482.
leukoaraiosis. Stroke 1999;30:478 – 479.
66. Dreher W, Kahn B, Gyngell ML, et al. Temporal and regional
91. Jones DK, Lythgoe D, Horsfield MA, et al. Characterization of
changes during focal ischemia in rat brain studied by proton
white matter damage in ischemic leukoaraiosis with diffusion
spectroscopic imaging and quantitative diffusion NMR imaging.
tensor MRI. Stroke 1999;30:393–397.
Magn Reson Med 1998;39:878 – 888.
92. Chabriat H, Vahedi K, Clark CA, et al. Decreased hemispheric
67. Ono J, Harada K, Mano T, Sakurai K, Okada S. Differentiation of
water mobility in hemiplegic migraine related to mutation of
dys- and demyelination using diffusional anisotropy. Pediatr Neu-
CACNA1A gene. Neurology 2000;54:510 –512.
rol 1997;16:63– 66.
93. Ford JC, Hackney DB, Lavi E, Phillips M, Patel U. Dependence of
68. Werring DJ, Clark CA, Barker GJ, Thompson AJ, Miller DH. Dif-
apparent diffusion coefficients on axonal spacing, membrane per-
fusion tensor imaging of lesions and normal-appearing white mat-
meability, and diffusion time in spinal cord white matter. J Magn
ter in multiple sclerosis. Neurology 1999;52:1626 –1632.
Reson Imaging 1998;8:775–782.
69. Tievsky AL, Ptak T, Farkas J. Investigation of apparent diffusion
94. Inglis BA, Yang L, Wirth ED, Plant D, Mareci TH. Diffusion anisot-
coefficient and diffusion tensor anisotropy in acute and chronic
multiple sclerosis lesions. AJNR 1999;20:1491–1499. ropy in excised normal rat spinal cord measured by NMR micros-
70. Iwasawa T, Matoba H, Ogi A, et al. Diffusion-weighted imaging of copy. Magn Reson Imaging 1997;15:441– 450.
the human optic nerve: a new approach to evaluate optic neuritis 95. Clark CA, Barker GJ, Tofts PS. Magnetic resonance diffusion im-
in multiple sclerosis. Magn Reson Med 1997;38:484 – 491. aging of the human cervical spinal cord in vivo. Magn Reson Med
71. Horsfield MA, Larsson HB, Jones DK, Gass A. Diffusion magnetic 1999;41:1269 –1273.
resonance imaging in multiple sclerosis. J Neurol Neurosurg Psy- 96. Clark CA, Werring DJ, Miller DH. Diffusion imaging in the spinal
chiatry 1998;64(suppl 1):S80 – 84. cord in vivo: estimation of the principal diffusivities and applica-
72. Ay H, Buonanno FS, Schaefer PW, et al. Posterior leukoencepha- tion to multiple sclerosis. Magn Reson Med 2000;43:133–138.
lopathy without severe hypertension: utility of diffusion-weighted 97. Rutherford MA, Cowan FM, Manzur AY. MR imaging of anisotro-
MRI. Neurology 1998;51:1369 –1376. pically restricted diffusion in the brain neonates and infants.
73. Hanyu H, Shindo H, Kakizaki D, et al. Increased water diffusion in J Comput Assist Tomogr 1991;15:188 –198.
cerebral white matter in Alzheimer’s disease. Gerontology 1997; 98. Toft PB, Leth H, Peitersen B, Lou HC, Thomsen C. The apparent
43:343–351. diffusion coefficient of water in gray and white matter of the infant
74. Hanyu H, Sakurai H, Iwamoto T, et al. Diffusion-weighted MR brain. J Comput Assist Tomogr 1996;20:1006 –1011.
imaging of the hippocampus and temporal white matter in Alzhei- 99. Vorisek I, Sykova E. Evolution of anisotropic diffusion in the de-
mer’s disease. J Neurol Sci 1998;156:195–200. veloping rat corpus callosum. J Neurophysiol 1997;78:912–919.
75. Chabriat H, Papatta S, Poupon C, et al. Clinical severity in CADA- 100. Prayer D, Roberts T, Barkovich AJ, et al. Diffusion-weighted MRI
SIL related to ultrastructural damage in white matter—in vivo of myelination in the rat brain following treatment with gonadal
study with diffusion tensor MRI. Stroke 1999;30:2637–2643. hormones. Neuroradiology 1997;39:320 –325.
76. Li TQ, Chen ZG, Hindmarsh T. Diffusion-weighted MR imaging of 101. Takeda K, Nomura Y, Sakuma H, et al. MR assessment of normal
acute cerebral ischemia. Acta Radiol 1998;39:460 – 473. brain development in neonates and infants: comparative study of
77. Baratti C, Barnett AS, Pierpaoli C. Comparative MR imaging study T1- and diffusion-weighted images. J Comput Assist Tomogr
of brain maturation in kittens with T1, T2, and the trace of the 1997;21:1–7.
diffusion tensor. Radiology 1999;210:133–142. 102. Beaulieu C, Fenrich FR, Allen PS. Multicomponent water proton
78. Zimmerman RA, Haselgrove JC, Wang ZY, et al. Advances in transverse relaxation and T2-discriminated water diffusion in my-
pediatric neuroimaging. Brain Dev 1998;20:275–289. elinated and nonmyelinated nerve. Magn Reson Imaging 1998;16:
79. Huppi PS, Maier SE, Peled S, et al. Microstructural development of 1201–1210.
human newborn cerebral white matter assessed in vivo by diffu- 103. Le Bihan D, Turner R, Douek P. Is water diffusion restricted in
sion tensor magnetic resonance imaging. Pediatr Res 1998;44: human brain white matter? An echo-planar NMR imaging study.
584 –590. Neuroreport 1993;4:887– 890.](https://image.slidesharecdn.com/dtibasics-110224131255-phpapp02/85/Dti-basics-12-320.jpg)
