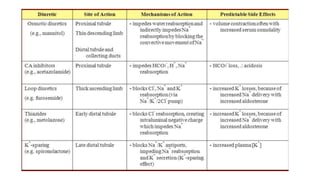
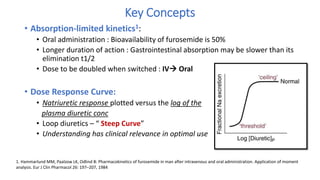
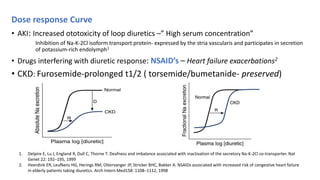
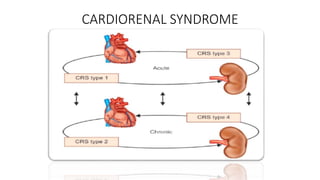
The document discusses the use of diuretics in various kidney diseases, including their pharmacology, clinical applications, and challenges such as diuretic resistance. It outlines different classes of diuretics, their mechanisms, and the importance of dosage adjustments based on patient conditions such as hypoalbuminemia and acute kidney injury (AKI). The conclusion emphasizes that while diuretics can be beneficial, their use needs careful evaluation in chronic kidney disease and AKI to avoid adverse outcomes.







![NEPHROTIC SYNDROME
• Renal albumin losses and reduced hepatic synthesis in the nephrotic syndrome
eventually lead to hypoalbuminemia
• Hypoalbuminemia reduces the binding of furosemide to plasma proteins and
thereby enlarges its volume of distribution1
• 2 studies have shown that patients with a serum albumin level of 2 g/dL can
deliver normal quantities of furosemide into the urine2
• Albumin infusion into nephrotic patients increased renal furosemide excretion,
whereas hypoalbuminemia enhances its metabolic clearance
• Agarwal et al3 found that displacing furosemide from albumin via
coadministration of sulfisoxazole did not affect natriuresis in nephrotic syndrome
1. Akcicek F, Yalniz T, Basci A, et al: Diuretic effect of frusemide in patients with nephrotic syndrome: is it potentiated
by intravenous albumin? [see comments]. Br Med J 310:162–163, 1995
2. Keller E, Hoppe-Seyler G, Schollmeyer P: Disposition and diuretic effect of furosemide in the nephrotic syndrome.
Clin Pharmacol Ther 32:442–449, 1982
3. Agarwal R, Gorski JC, Sundblad K, et al: Urinary protein binding does not affect response to furosemide in patients
with nephrotic syndrome. J Am Soc Nephrol 11:1100–1105, 2000](https://image.slidesharecdn.com/diureticsppt-190922155951/85/Diuretics-ppt-11-320.jpg)






![• Loop diuretics :Increase the risk of cast formation in crush syndrome by acidifying
the urine1 and worsen already existing hypocalcemia by inducing hypercalciuria
• Recommend: Do not use loop diuretics routinely for prevention or treatment of
AKI2
1. Better OS, Stein JH. Early management of shock and prophylaxis of acute renal failure in
traumatic rhabdomyolysis. N Engl J Med 1990; 322: 825–829
2. KDIGO Acute Kidney Injury Work Group. KDIGO Clinical Practice Guidelines for Acute Kidney
Injury. Kidney Intl 2012; 2[Suppl]: 1–138](https://image.slidesharecdn.com/diureticsppt-190922155951/85/Diuretics-ppt-18-320.jpg)













![• Results: Bolus with continuous infusion,
• There was NO significant difference in patients’ global assessment of
symptoms (mean AUC, 4236±1440 and 4373±1404, respectively; P =
0.47) or in the mean change in the creatinine level (0.05±0.3 mg per
deciliter [4.4±26.5 μmol per liter] and 0.07±0.3 mg per deciliter
[6.2±26.5 μmol per liter], respectively; P = 0.45)
• Conclusion : NO significant differences in patients’ global assessment of
symptoms or in the change in renal function when diuretic therapy was
administered by bolus as compared with continuous infusion or at a high
dose as compared with a low dose
DOSE
TRIAL
2011](https://image.slidesharecdn.com/diureticsppt-190922155951/85/Diuretics-ppt-33-320.jpg)

![RCTs on UF Vs Diuretics in HF
Reference Study Design and
protocol
Patients (n) Ultrafiltration Therapy Effect on
Renal Function
Main Findings
Hanna (2012) Patients with NYHA
class III/IV heart
failure
and ejection fraction
,40%,UF rate 400
ml/h for 6 h and 200
ml/h thereafter
36 UF group had a fluid
removal rate of 3.4
ml/kg per hour with
a total volume
removal of 5.2 L. The
mean time to
achieve a PCWP of
#18 mmHg was 22 h
in the UF group
No significant
difference in serum
creatinine and
cystatin C levels
before and after
therapy between the
two groups
Fluid removal was
faster and more
efficient in the UF
group with shorter
hospital length of
stay, no change in
inflammatory
markers, NT-pro-
BNP, hospital
readmissions
Bart (2012)
[CARRESS-HF]
UF was used as
rescue
therapy after
patients
had worsening renal
function (UF rate
200 ml/h)
188 Median duration of
UF was 40 h. Post-UF
weight loss was 5.7
kg
Serum creatinine
level increased
significantly after UF.
No change in serum
creatinine level with
medical Therapy
Although weight loss
was similar with UF
and pharmacologic
therapy, patients in
the UF group had
higher rate of
serious adverse
events.
9/22/2019 35](https://image.slidesharecdn.com/diureticsppt-190922155951/85/Diuretics-ppt-35-320.jpg)








