Diuretics in Heart Failure: Key Trials
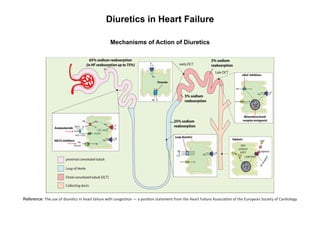
- 1. Diuretics in Heart Failure Mechanisms of Action of Diuretics Reference: The use of diuretics in heart failure with congestion — a position statement from the Heart Failure Association of the European Society of Cardiology
- 4. THIAZIDES
- 5. K+-SPARING AGENTS Reference: KAPLAN Medical USMLE Step 1 Lecture Note 2018 Pharmacology
- 6. Reference: ESC Guidelines of Heart Failure
- 7. Reference: Bonow RO, Mann DL, Zipes DP, Libby P. Braunwald’s Heart Disease e-Book: A Textbook of Cardiovascular Medicine.; 2020.
- 8. Flowchart to Diuretic use in Acute Heart Failure (A) Congestion with volume overload. (B) Treatment algorithm after 24 h. Total loop diuretic dose can be administered either as continuous infusion or bolus infusion. BP, blood pressure; HF, heart failure; IV, intravenous; SGLT2‐I, sodium–glucose linked transporter 2 inhibitor; UF, ultrafiltration; UO, urine output. Higher dose should be considered in patients with reduced glomerular filtration rate. *Consider other reasons for dyspnoea given the quick resolution of congestion. °The maximal dose for IV loop diuretics is generally considered furosemide 400–600 mg or 10–15 mg bumetanide. In patients with good diuresis following a single loop diuretic administration, once a day dosing can be considered. Reference: European Journal of Heart Failure / Volume 21, Issue 2 / p. 137-155
- 13. Reference: ESC Guidelines Diagnosis and Treatments Acute and Chronic Heart Failure Supplementary Data
- 14. Algoritma Tatalaksana Resistensi Diuretik Reference: Journal of the American College of Cardiology Vol. 75, NO. 10, 2020 ª 2020 By the American College of Cardiology Foundation Published by Elsevier
- 15. Trials in Heart Failure TRANSFORM-HF The TRANSFORM-HF trial failed to show that torsemide was superior to furosemide in improving survival among patients treated for decompensated heart failure. Primary outcome: All-cause mortality, occurred in 26.2% of the furosemide group vs. 26.1% of the torsemide group (p = 0.77). Secondary outcomes: All-cause mortality or hospitalization: 49.3% of the furosemide group vs. 47.3% of the torsemide group (p = 0.11) Conclusions: • Among patients admitted with decompensated heart failure, post-discharge management with torsemide was not superior to furosemide. • All-cause mortality was similar between treatment groups. • Either furosemide or torsemide are acceptable options for diuresis among patients with heart failure. Reference: Verbrugge Frederik H, Menon Venu. European Heart Journal: Acute Cardiovascular Care (2022) 11, 931–932. Diuretic Optimization Strategies Evaluation in Acute Heart Failure – DOSE HF The DOSE study sought to evaluate the safety and efficacy of two strategies for furosemide dosing in patients with ADHF: 1) route of administration (by means of either a bolus every 12 hours or continuous infusion) 2) dosing (at either a low dose (equivalent to the patient’s previous oral dose) or a high dose (2.5 times the previous oral dose) The coprimary end points: Patients’ global assessment of symptoms, quantified as the area under the curve (AUC) of the score on a visual-analogue scale over the course of 72 hours, and the change in the serum creatinine level from baseline to 72 hours.
- 16. Conclusion: Among patients with acute decompensated heart failure, there were no significant differences in patients’ global assessment of symptoms or in the change in renal function when diuretic therapy was administered by bolus as compared with continuous infusion or at a high dose as compared with a low dose. Reference: Felker GM, Lee KL, Bull DA, et al., on behalf of the NHLBI Heart Failure Clinical Research Network. Diuretic strategies in patients with acute decompensated heart failure. N Engl J Med 2011;364:797-805. Cardiorenal Rescue Study in Acute Decompensated Heart Failure - CARRESS-HF The current trial sought to investigate if Slow Continuous Ultra Filtration (SCUF) would be superior to stepped pharmacological treatment (conventional management) in improving renal function and promoting weight loss/diuresis in patients admitted with ADHF with evidence of worsening renal function. Primary Endpoints: Change in serum Cr and change in weight between randomization and 96 hours, considered as a bivariate response Conclusions: • The results of the CARRESS-CHF trial indicate that SCUF is not superior to stepped pharmacological treatment in the management of patients with ADHF who have cardiorenal syndrome. • Serum Cr increased rather than decreased in the SCUF arm by 48 hours. • SCUF also resulted in worsening hyponatremia and a higher incidence of adverse events including some related to SCUF administration. Reference: Bart BA, Goldsmith SR, Lee KL, et al. Ultrafiltration in decompensated heart failure with cardiorenal syndrome. N Engl J Med 2012;367:2296-2304. Renal Optimization Strategies Evaluation in Acute Heart Failure - ROSE AHF The goal of the trial was to evaluate treatment with low-dose dopamine or low-dose nesiritide compared with placebo among patients with acute heart failure and renal dysfunction.
- 17. Primary Endpoints: • Cumulative urinary volume at 72 hours • Change in cystatin-C at 72 hours Conclusions: • Among patients with acute heart failure and renal dysfunction, neither low-dose dopamine nor low-dose nesiritide enhanced decongestion or improved renal function. • These findings differ from results of previous small studies. • Nesiritide and dopamine are used in a significant proportion of heart failure patients, and in the case of dopamine, recommended by heart failure guidelines. • The use of these agents will likely need to be re-evaluated in the setting of acute heart failure and renal dysfunction. Reference: Chen HH, Anstrom KJ, Givertz MM, et al., on behalf of the NHLBI Heart Failure Clinical Research Network. Low-Dose Dopamine or Low-Dose Nesiritide in Acute Heart Failure with Renal Dysfunction: The ROSE Acute Heart Failure Randomized Trial. JAMA 2013;310:2533-43. Comparison of Metolazone Versus Chlorothiazide in Acute Decompensated Heart Failure with Diuretic Resistance The goal of the trial was to evaluate the efficacy and safety of oral metolazone versus IV chlorothiazide as add-on therapy to loop diuretics in patients hospitalized with ADHF and renal dysfunction. Primary endpoint: Net urine output (UOP) at 72 h after initiation of thiazide-like diuretics. Conclusion: Sequential nephron blockade with either metolazone or chlorothiazide appears to be efficacious and safe in ADHF, renal dysfunction, and diuretic resistance. Reference: P. Moranville Michael, Choi Suji, et al. Comparison of Metolazone Versus Chlorothiazide in Acute Decompensated Heart Failure with Diuretic Resistance. Cardiovascular Therapeutics 33 (2015) 42–49.
- 18. Aldosterone Targeted NeuroHormonal Combined with Natriuresis Therapy – Heart Failure - ATHENA-HF The goal of the trial was to evaluate treatment with a high-dose mineralocorticoid receptor antagonist (MRA) compared with placebo among patients admitted with acute heart failure. Primary endpoint: To assess the effect of high-dose spironolactone and usual care on N-terminal pro-B-type natriuretic peptide (NT-proBNP) levels compared with usual care alone. Conclusions: • Among patients admitted with acute heart failure, high-dose spironolactone was not effective at reducing NT-proBNP levels. • Secondary outcomes, including dyspnea, urine output, and change in potassium, were also similar between treatment groups. Reference: Efficacy and Safety of Spironolactone in Acute Heart Failure: The ATHENA-HF Randomized Clinical Trial. JAMA Cardiol 2017;Jul 12:[Epub ahead of print]. Acetazolamide in Decompensated Heart Failure with Volume Overload – ADVOR The goal of the trial was to compare incidence of successful decongestion among patients with acute decompensated heart failure receiving intravenous (IV) loop diuretics with IV acetazolamide versus placebo. Primary endpoint: Successful decongestion, defined as the absence of signs of volume overload, within 3 days after randomization and without an indication for escalation of decongestive therapy. Conclusion: The addition of acetazolamide to loop diuretic therapy in patients with acute decompensated heart failure resulted in a greater incidence of successful decongestion. References: Mullens W, Dauw J, Martens P, et al., on behalf of the ADVOR Study Group. Acetazolamide in Acute Decompensated Heart Failure with Volume Overload. N Engl J Med 2022;387:1185-95.
- 19. Efficacy of Vasopressin Antagonism in Heart Failure: Outcome Study with Tolvaptan – EVEREST The goal of the trial was to evaluate the safety and efficacy of tolvaptan compared with placebo among patients hospitalized with acute decompensated heart failure (ADHF). Primary endpoint: • All-cause mortality • CV mortality or heart failure hospitalization Conclusion: Tolvaptan initiated for acute treatment of patients hospitalized with heart failure had no effect on long-term mortality or heart failure–related morbidity Reference: Konstam MA, Gheorghiade M, Burnett JC Jr, et al. Effects of oral tolvaptan in patients hospitalized for worsening heart failure: the EVEREST Outcome Trial. JAMA 2007;297:1319-31.