The document discusses chemical reactions and provides examples to illustrate different types of chemical reactions:
1) Combination reactions involve reactants combining to form a single product, like burning of coal or formation of water.
2) Decomposition reactions involve a single reactant breaking down into simpler products, like heating ferrous sulfate crystals.
3) Displacement reactions involve one element displacing another from a compound, like an iron nail causing copper ions to displace from copper sulfate solution.
4) Precipitation reactions form an insoluble product or precipitate, like mixing sodium sulfate and barium chloride solutions.
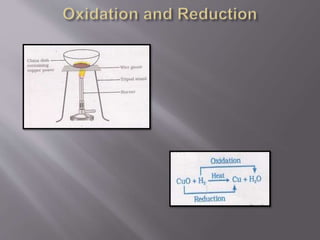
5) Oxidation-reduction reactions involve transfer of electrons leading to oxidation and reduction of reactants.









![Let us understand it by a activity :-
o Take about 2gm ferrous sulphate crystals in a dry boiling tube.
o Note the color of the ferrous sulphate crystals.
o Heat the boiling tube over the flame or a burner or spirit lamp as
shown.
o Observe the color of the crystals after heating.
Have you noticed that the green color of the ferrous sulphate crystals has
changed? You can also smell the characteristic odor of burning sulphur.
2FeSO4(s) Heat---------- Fe2O3(s) + SO2(g) + SO3(g)
{Ferrous Sulphate} [ Ferric Oxide ]
In this reaction you can observe that a single reactant breaks down to give
simpler products. This is a decomposition reaction. Ferrous sulphate
crystals (FeSO4, 7H2O) lose water when heated and the color of the
crystals changes. Or then decomposes to ferric oxide, sulphur dioxide and
sulphur trioxide. Ferric oxide is a solid, while SO2 and SO3 are gases.](https://image.slidesharecdn.com/chemicalreactionandequations-150623114507-lva1-app6892/85/Chemical-reaction-and-equations-10-320.jpg)