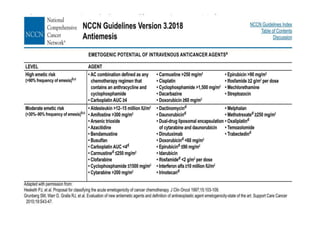
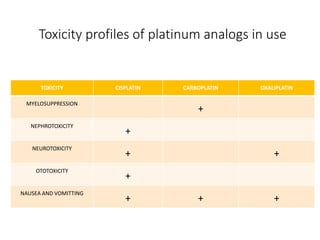
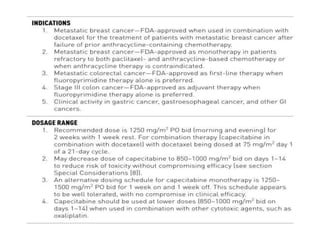
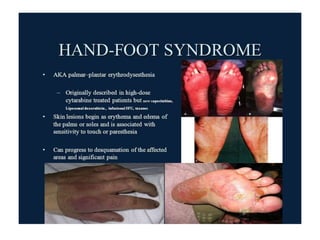
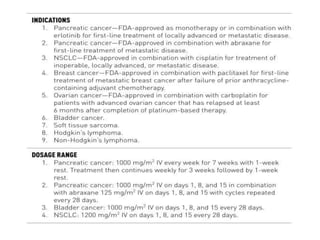
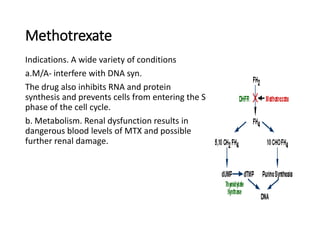
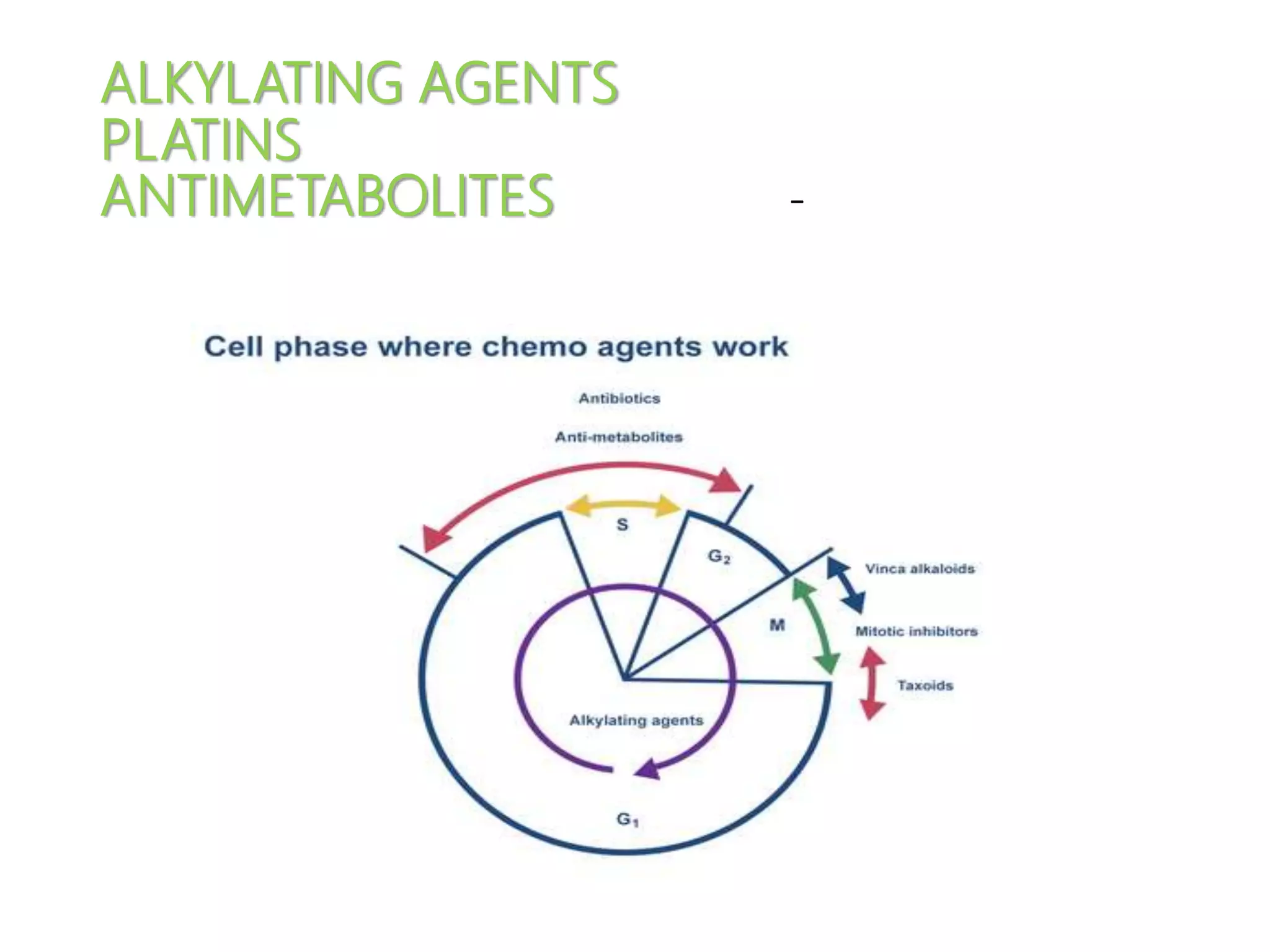
This document summarizes several alkylating agents used in chemotherapy. Alkylating agents work by alkylating DNA and RNA, which can cause DNA breaks or abnormal sequences and damage cells' ability to replicate. Common side effects include myelosuppression, nausea/vomiting, and alopecia. Specific agents discussed include cyclophosphamide, ifosfamide, melphalan, busulfan, carmustine, lomustine, procarbazine, dacarbazine, streptozocin, bendamustine, altretamine, and chlorambucil. Each has unique indications and toxicity profiles involving bone marrow suppression, gastrointestinal upset, organ toxicity, secondary cancers, and more.














![Carmustine [BCNU, bischlorethyl nitrosourea (BiCNU)]
Indications. Brain tumors, myeloma, HL & NHL. In high doses for BMT.
In the form of implantable wafers: GBM
Pharmacology
a. M/A. Forms interstrand cross-links in DNA preventing DNA replication and
transcription.
b. Metabolism. Highly lipid-soluble drug that enters the brain. Rapid spontaneous
decomposition to active and inert product. Most of the intact drug and metabolic
products are excreted in urine.
Toxicity
a. Dose limiting. Myelosuppression ,aggravated by concurrent RT.
b. Common. Nausea and vomiting. Local pain during injection.
c. Occasional. Stomatitis, esophagitis, diarrhea, LFT abnormalities; alopecia, facial
flushing, brown discoloration of skin; interstitial lung disease with pulmonary
fibrosis ,dizziness, optic neuritis, ataxia, organic brain syndrome; renal insufficiency
d. Rare. 2nd cancer
Lomustine [CCNU, cyclohexyl chlorethyl nitrosourea]
Indications. Brain tumors & HL](https://image.slidesharecdn.com/chapter21alkylatingagentsplatinsantimetabolites-191105205002/85/Chapter-21-alkylating-agents-platins-antimetabolites-16-320.jpg)