The key factors that can influence toxicity are:
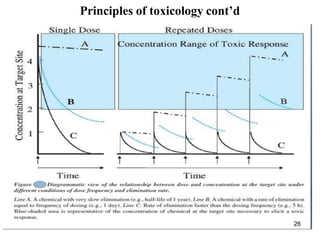
1. Quantity of toxin - A higher dose will generally cause more severe effects than a lower dose.
2. Route of exposure - Different routes like ingestion, inhalation, injection affect toxicity. Inhalation/injection often more toxic.
3. Individual susceptibility - Factors like age, health status, genetics can impact susceptibility. The very young and old often more vulnerable.
4. Type of toxin - Properties like solubility, ability to accumulate, interact with other chemicals impact toxicity.
5. Duration of exposure - Long-term/repeated exposure often more toxic than short-term exposure due to accumulation over time.








































































![Initial management of the poisoned patient…
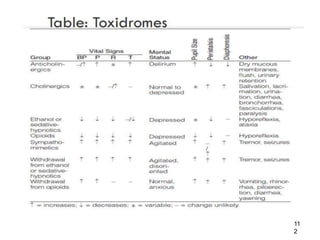
g) Hyperthermia:
May be associated with poisoning by amphetamines (esp methylene dioxy
methamphetamine [MDMA; “Ecstasy”]), atropine & other anti-cholinergic
drugs, cocaine, salicylates, strychnine, TCAs, & various other medications
Overdoses of SSRIs or antipsychotic agents can cause rigidity &
hyperthermia
79](https://image.slidesharecdn.com/chapter1introduction-240306162850-e711f858/85/chapter-1-introduction-for-health-pptx-79-320.jpg)



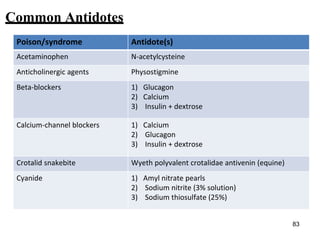
![Common antidotes…
Poison/syndrome Antidote(s)
Digitalis Digoxin immune Fab (Digibind)
Ethylene glycol
Methanol
1) Ethanol 10% in D5W ± hemodialysis
2) Fomepizole [4-MP] (Antizol) ± hemodialysis
Heparin Protamine sulfate
Hydrofluoric Acid Calcium gluconate
Iron Deferoxamine
Isoniazid Pyridoxine (Vitamin B6)
Lead 2,3-dimercaptosuccinic acid [DMSA] (Succimer)
Mercury
Arsenic
Gold
British antilewisite, dimercaprol (BAL); in peanut oil
84](https://image.slidesharecdn.com/chapter1introduction-240306162850-e711f858/85/chapter-1-introduction-for-health-pptx-84-320.jpg)
![Common Antidotes…
Poison/syndrome Antidote(s)
Carbon monoxide Oxygen ± hyperbaric chamber
Methemoglobinemia Methylene blue (1 percent solution)
Opiates Naloxone
Nalmefene, naltrexone
Organophosphates
Carbamates
Nerve agents
1) Atropine
2) Pralidoxime [2-PAM]
Sulfonylurea Octreotide (Sandostatin) + dextrose
Benzodiazepines Flumazenil
Tricyclic
antidepressants
Sodium bicarbonate (NaHCO3)
85](https://image.slidesharecdn.com/chapter1introduction-240306162850-e711f858/85/chapter-1-introduction-for-health-pptx-85-320.jpg)