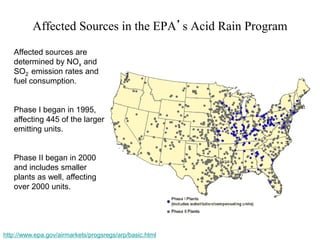
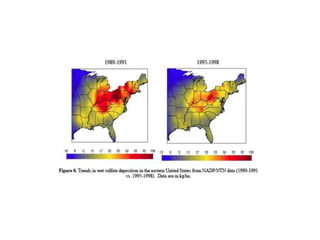
Acid rain is caused by emissions of sulfur and nitrogen oxides from fossil fuel combustion. It damages sensitive ecosystems through acidification of soils and freshwater bodies. The key effects are leaching of nutrients from soils, release of aluminum which is toxic to plants and fish, and loss of biodiversity in freshwater systems. The Clean Air Act established a two-phase program in the 1990s that significantly reduced sulfur dioxide emissions from power plants in the eastern U.S., achieving over a 50% reduction in related acid rain. However, developing accurate source-receptor models to guide regulation remains an ongoing challenge.