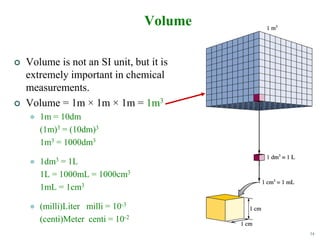
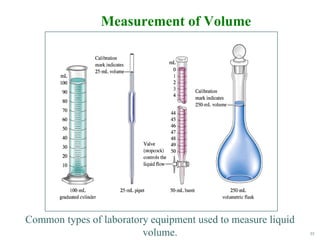
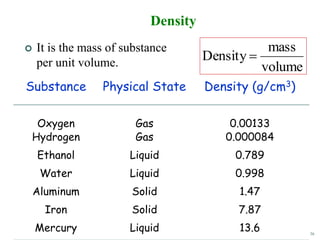
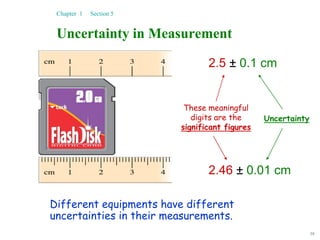
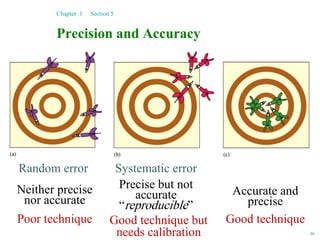
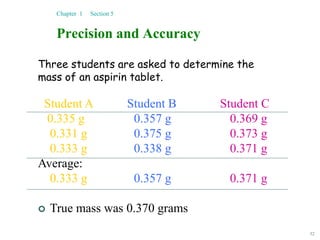
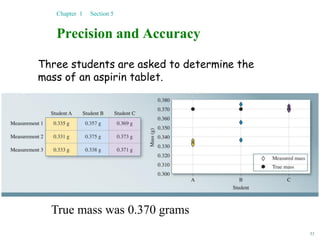
This document discusses several key concepts in chemistry including the following:
1. Everything in the universe is made of approximately 100 different types of atoms that combine to form all substances. Chemical reactions involving these atoms are important for life and cause many problems.
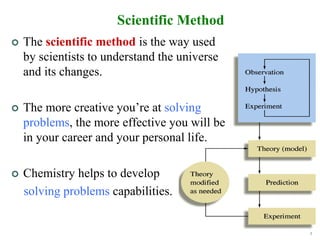
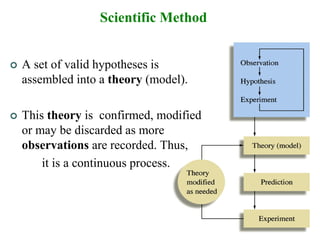
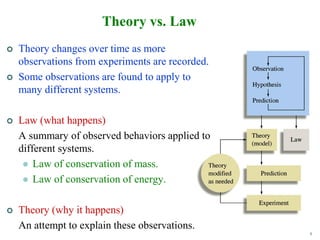
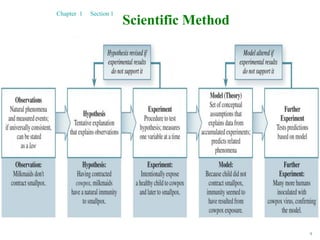
2. The scientific method is used by scientists to understand the universe through observation, hypothesis, experimentation, and developing theories. Theories can be modified over time with new evidence.
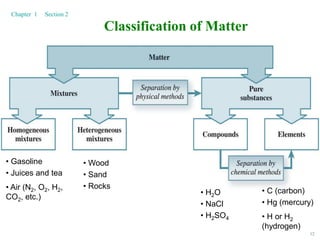
3. Matter can exist as elements, compounds, mixtures, gases, liquids, and solids. Various separation and classification methods are used to distinguish these forms of matter based on their physical and chemical properties.






















![32
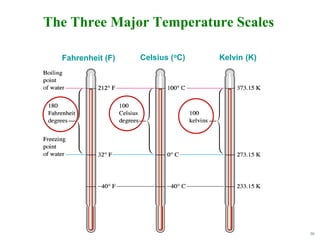
Fahrenheit Scale vs. Celsius Scale
Both unit temperature
size and zero locations
are different.
Since:
180°F = 100°C => 9°F =
5°C
and:
32°F = 0°C
Then to convert from °F to °C:
[Tf (°F) – 32 (°F)] = Tc
(°C)
5°C
9°F](https://image.slidesharecdn.com/ch1-230806192849-9bf30f55/85/CH1-pdf-32-320.jpg)
![33
Fahrenheit Scale vs. Celsius Scale
[ Tc (a) – Tc (b) ] °C = (5°C/9°F) [ Tf (a) – Tf (b) ] °F
When Tc (b) = - 40°C, then Tf (b) = -40°F
giving that:
[ Tc (a) – (-40) ] °C = (5°C/9°F) [ Tf (a) – (-40) ] °F
Thus:
or
F
9
C
5
40
40
f
c
T
T
C
5
F
9
40
40
c
f
T
T](https://image.slidesharecdn.com/ch1-230806192849-9bf30f55/85/CH1-pdf-33-320.jpg)