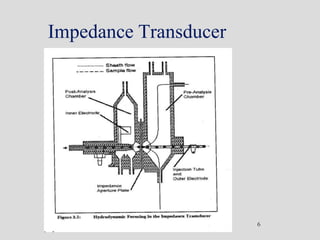
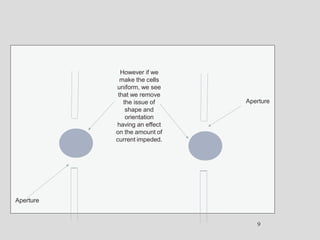
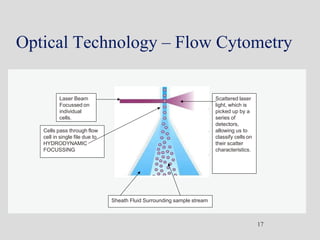
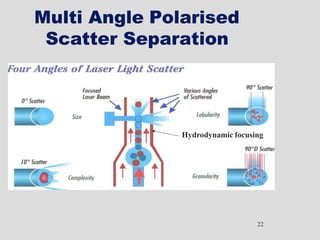
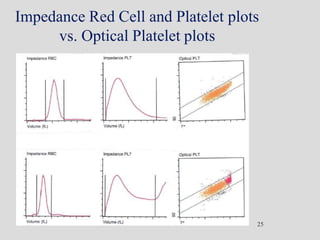
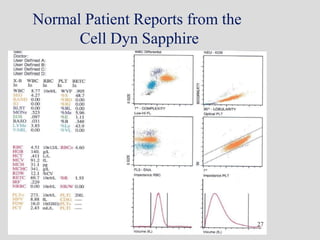
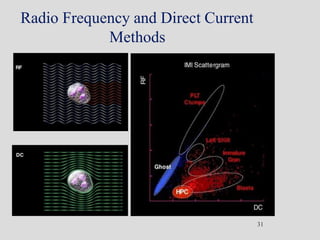
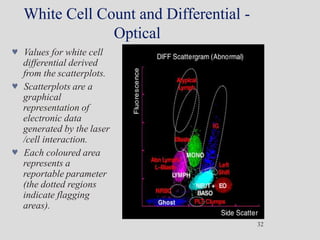
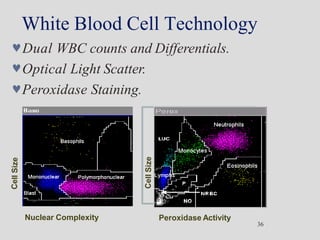
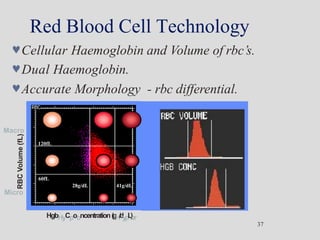
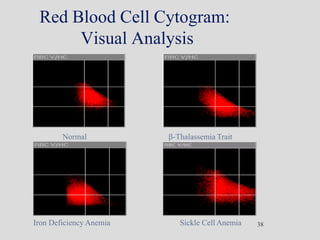
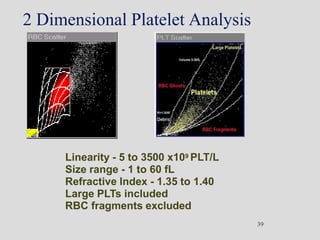
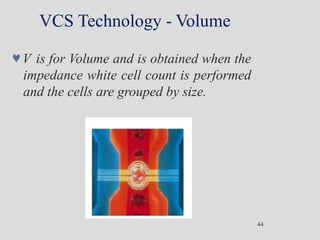
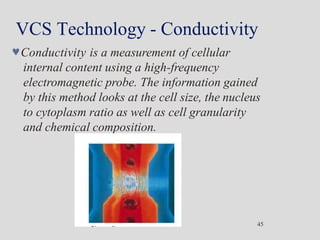
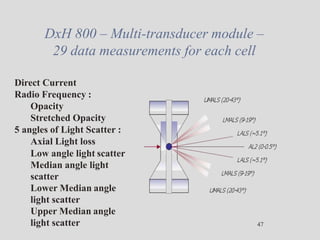
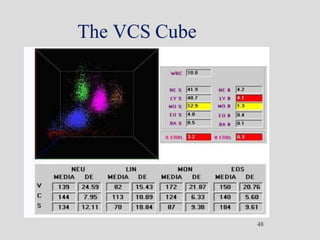
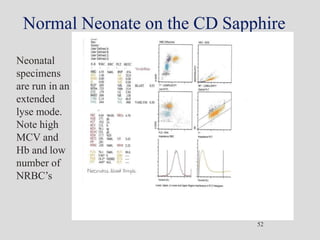
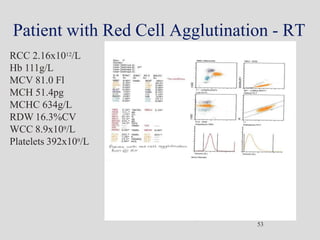
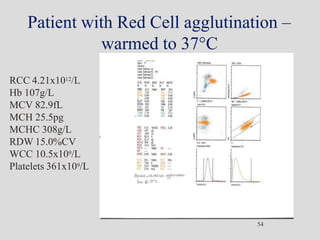
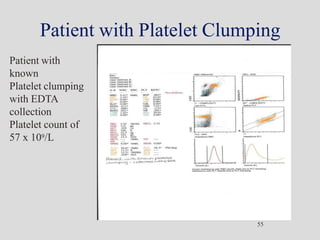
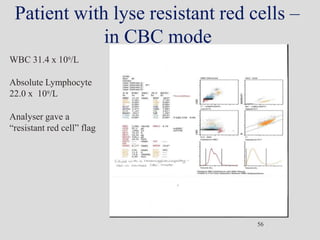
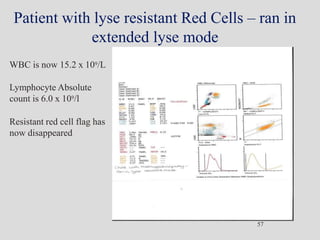
Wallace Coulter developed the Coulter Principle for counting and sizing blood cells using impedance technology in the late 1940s. Modern cell counters use various technologies including impedance, absorbance spectrophotometry, optical light scattering, fluorescence, radio frequency, and monoclonal antibodies to provide complete blood cell counts, differentials, reticulocyte counts, and other parameters from small blood samples. Cell counters must operate within quality control standards to provide accurate and precise results.