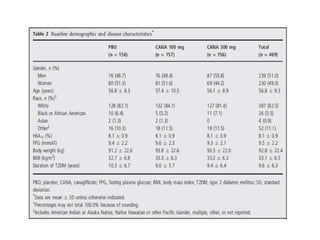
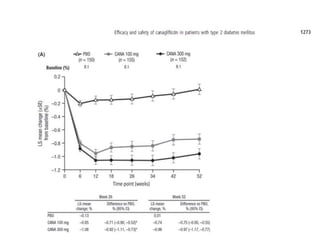
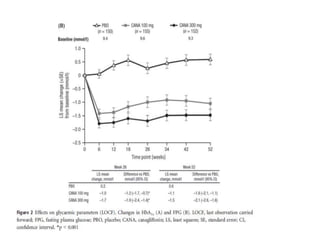
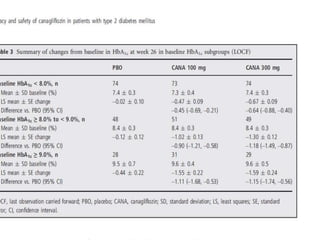
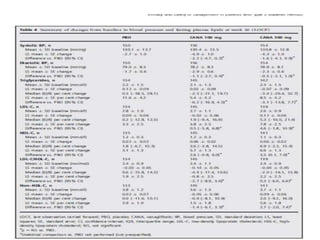
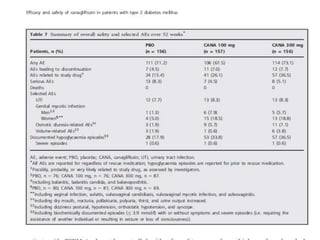
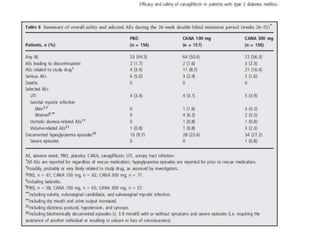
This randomized controlled trial evaluated the efficacy and safety of canagliflozin compared to placebo in patients with type 2 diabetes inadequately controlled with metformin and sulfonylurea. At 26 weeks, HbA1c and fasting plasma glucose were significantly reduced from baseline with canagliflozin 100mg and 300mg compared to placebo. Reductions were maintained at 52 weeks. Canagliflozin also reduced body weight and systolic blood pressure more than placebo. Overall adverse event rates were similar, but genital mycotic infections and osmotic diuresis events were more common with canagliflozin. Increased non-severe hypoglycemia was also seen with canagliflozin.