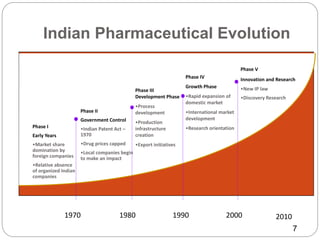
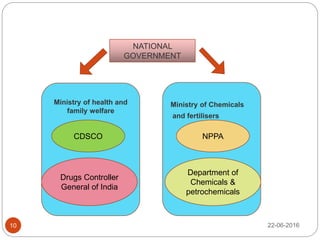
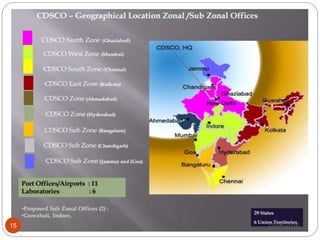
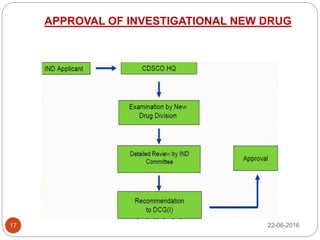
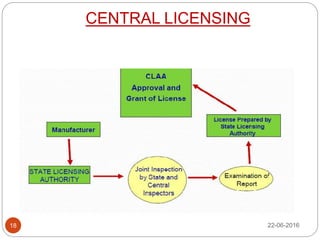
This document provides an overview of the major regulatory bodies that govern the Indian pharmaceutical sector, with a focus on the Central Drugs Standard Control Organization (CDSCO). It begins with background on drug regulation and the need for effective regulation. It then discusses various international and Indian regulatory bodies. The bulk of the document describes the roles and functions of CDSCO and the Drug Controller General of India as the central drug authorities that approve clinical trials, marketing authorization, and licenses for certain drug categories. It also briefly discusses the National Pharmaceutical Pricing Authority and deficiencies in India's drug regulatory system.