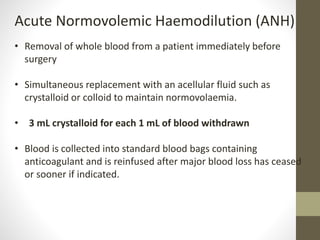
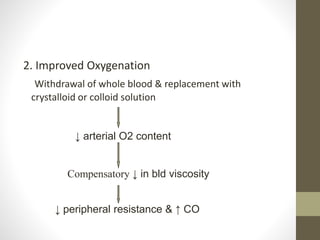
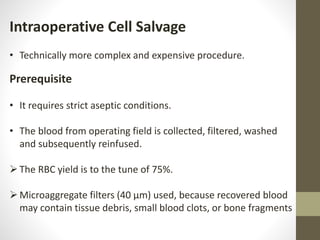
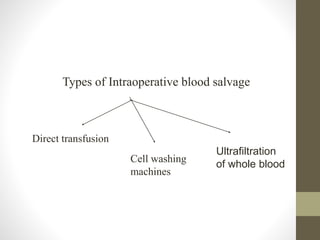
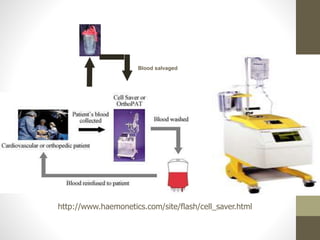
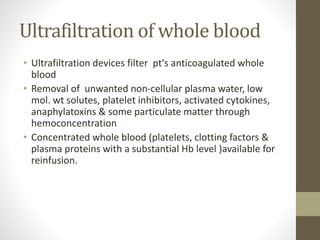
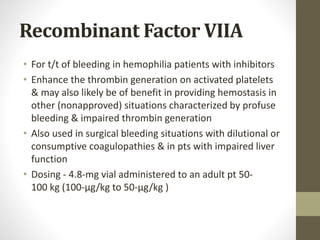
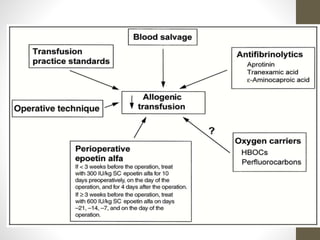
This document discusses blood conservation techniques used in the perioperative period. It begins by defining blood conservation and appropriate transfusion. It then lists guiding principles, including that transfusions carry risks and conservation provides alternatives. It discusses autologous versus allogeneic transfusions. Techniques discussed include preoperative autologous donation, intraoperative methods like positioning, regional anesthesia, deliberate hypotension, and pharmacological agents. Intraoperative blood salvage and postoperative salvage are also mentioned.