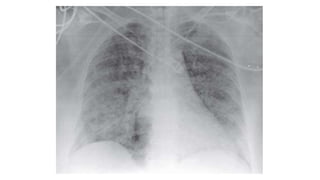
ARDS-acute respiratory distress syndrome
- 2. Ddx • Cardiogenic pulmonary Edema • ARDS • Bilateral pneumonia • Alveolar haemorrhage • Acute interstitial lung diseases (e.g., acute interstitial pneumonitis) • Acute immunologic injury (e.g., hypersensitivity Pneumonitis) • Toxin injury (e.g., radiation pneumonitis)
- 4. What is ARDS? • As per the Berlin definition: 1. Onset: Acute-Within 1 week of clinical insult or new or worsening respiratory symptoms 2. Chest Imaging (Xray or CT): Bilateral opacities not fully explained by effusions, lobar/lung collapse, or Nodules 3. Origin of Edema: Respiratory failure not fully explained by cardiac failure or fluid overload with objective assessment (eg: ECHO, BNP, Cardiac Catheterization) to exclude hydrostatic edema if no risk factor is present.
- 5. What is ARDS? 4. Hypoxemia • Mild: PaO2/FiO2 200-300 • Moderate: PaO2/FiO2 100-200 • Severe: PaO2/FiO2 ≤100 ** with PEEP or CPAP ≥ 5cm H2O
- 6. Etiology • Can be caused by both pulmonary and non pulmonary causes like: • DIRECT LUNG INJURY INDIRECT LUNG INJURY Pneumonia Sepsis Aspiration of gastric contents Pancreatitis Pulmonary contusion Multiple transfusions Near-drowning Burns Toxic inhalation injury Severe trauma Fat embolism
- 7. Risk Factors • There are a variety of risk factors, including • Alcohol abuse • Obesity • Patients who present with Acute Physiology and Chronic Health Evaluation (APACHE) scores > 16 • Increased use of transfusions • Septic shock • Chronic conditions like DM, HTN, Malignancy, Immunocompromised states, CLD, CKD etc.
- 8. Pathogenesis
- 9. Pathogenesis • 3 phases-Exudative, Proliferative, Fibrotic • Exudative Phase: • Diffuse Alveolar Damage/Capillary endothelial damage • Type 1 Pneumocytes/Endothelial cells • Loss of tight junctions • Inflammatory mediators release • Neutrophilic infiltration • Leakage of protein rich fluid + Inflammatory cells leading to • Edema and Hyaline membrane formation – Hallmark of ARDS
- 11. Pathogenesis • Exudative Phase • Alveolar edema mainly involves dependent portions • Collapse of large segments of lung leading to intrapulmonary shunting and hypoxemia • To compensate, work of breathing increases leading to dypnoea • There is a concurrent injury to pulmonary vasculature as well • So, even in normal areas of lung, reduced arterial blood flow leads to increased dead space and hypercapnia
- 12. Pathogenesis • Proliferative Phase: • First signs of resolution are often evident in this phase, with the initiation of lung repair • Organization of alveolar exudates, and a shift from neutrophil- to lymphocyte-predominant pulmonary infiltrates. • Type II pneumocytes proliferate • Synthesize new pulmonary surfactant and differentiate into type I pneumocytes.
- 13. Pathogenesis • Fibrotic Phase: • Not all patient progress to the fibrotic stage. Mechanism not well understood. • Alveolar edema and inflammatory exudates of earlier phases convert to extensive alveolar-duct and interstitial fibrosis. • Marked disruption of acinar architecture leads to emphysema-like changes, with large bullae. • Intimal fibroproliferation in the pulmonary microcirculation causes progressive vascular occlusion and pulmonary hypertension. • Consequently there is an increased risk of pneumothorax, reduction in lung compliance, and increased pulmonary dead space.
- 14. Timeline
- 15. Clinical Feature • Dyspnea • Cyanosis • Diffuse crepitations • Tachypnea • Tachycardia • Use of Acc. Muscles of respiration • ABG-Hypoxemia, Increased A-a O2 gradient
- 16. Treatment • Main focus is on treating the underlying pathology with supportive management which requires: • Maintaining the Airway • Oxygenation • Supporting hemodynamic function. • The five P's of supportive therapy include • Protective lung ventilation, • Perfusion, • Positioning, • Preventing complications • Protocol weaning
- 18. Stepwise management principles • Strategies to avoid invasive ventilation • Lung protective invasive ventilation • Rescue strategies for hypoxia: • Recruitment Manouvers • Prone positioning • HFOV/APRV • ECMO • Inhaled NO/Prostaglandins
- 19. Advantages of HFNC • Hgiher Fio2 delivery (21%-100%) • Reduction in anatomical dead space • Reduces work of breathing • PEEP generation which causes recruitment • Delivery of warmed and humidified gases
- 20. Predictors of HFNC failure • ROX index= 𝑆𝑝𝑜2 𝐹𝑖𝑂2 𝑅𝑒𝑠𝑝𝑖𝑟𝑎𝑡𝑜𝑟𝑦 𝑅𝑎𝑡𝑒 • Essentially if Spo2 is falling OR Higher FiO2 is needed or Patient’s RR is increasing s/o NIV failure • HFNC (after 2 hrs)=ROX index <2.85 • HFNC (after 6 hrs or 12 hrs)=ROX index <3.85
- 22. HFNC Failed … Now What?
- 23. NIV (BiPAP/CPAP) • HACOR SCORE • Bedside predictor of NIV failure • HACOR Score>5 after 1 hr of NIV = 80% failure of NIV regardless of disease severity, Diagnosis, Age.
- 25. Why so many predictors for NIV are being used? Can’t we just wait and watch?
- 26. No
- 27. Why not? Late intubation and late use of mechanical ventilation is associated with increased mortality!
- 29. Why the need for mechanical ventilation? • Most patients of ARDS require mechanical ventilation as they frequently become fatigued from increased work of breathing and progressive hypoxemia, requiring mechanical ventilation for support.
- 30. Mechanical Ventilation-A double edged sword • Can lead to Ventilator -Induced Lung Injury Despite its lifesaving potential • Arises from three principal mechanisms (explain): • Volutrauma • Atelectrauma • Biotrauma-High FiO2>65% aggravates Alveolar damage due to oxygen toxicity
- 31. Solution? • LUNG PROTECTIVE VENTILATION aka Low VT ventilation (4 to 8 mL/Kg of PBW. Usually 6 mL/kg of predicted body weight) • PBW=50 or 45.5 + 2.3*(ht in inches – 60) • Plateau pressure ≤30 cm H2O • FiO2 target <65% • Provide PEEP(Low vs High->12cm H2O) • Find the best PEEP by titrate with respect to PaO2, FiO2 • Aim is to prevent alveolar collapse and also prevent over distention • PEEP<<CPAP • Permissive Hypercapnia (Increase RR, Bicarb) • SpO2 target =85 to 90%
- 32. Step by step ventilator management PART I: VENTILATOR SETUP AND ADJUSTMENT 1. Calculate predicted body weight (PBW) 2. Select any ventilator mode as per clinical discretion 3. Set ventilator settings to achieve initial VT = 8 ml/kg PBW 4. Reduce VT by 1 ml/kg at intervals ≤ 2 hours until VT = 6ml/kg PBW. 5. Set initial rate to approximate baseline minute ventilation (not > 35 bpm). 6. Adjust VT and RR to achieve pH, oxygenation and plateau pressure goals as mentioned.
- 33. Step by step ventilator management • OXYGENATION GOAL: PaO2 55-80 mmHg or SpO2 88-95% • Use a minimum PEEP of 5 cm H2O. Consider use of incremental FiO2/PEEP combinations such as shown below (not required) to achieve goal.
- 34. Step by step ventilator management PLATEAU PRESSURE GOAL: ≤ 30 cm H2O Check Pplat (0.5 second inspiratory pause), at least q 4h and after each change in PEEP or VT. • If Pplat > 30 cm H2O: decrease VT by 1ml/kg steps (minimum = 4 ml/kg). • If Pplat < 25 cm H2O and VT< 6 ml/kg, increase VT by 1 ml/kg until • Pplat > 25 cm H2O or VT = 6 ml/kg. • If Pplat < 30 and dys-synchrony occurs: may increase VT in 1ml/kg increments to 7 or 8 ml/kg if Pplat remains < 30 cm H2O.
- 35. Step by step ventilator management pH GOAL: 7.30-7.45 Acidosis Management: (pH < 7.30) • If pH 7.15-7.30: Increase RR until pH > 7.30 or PaCO2 < 25 (Max. set RR = 35). • If pH < 7.15: Increase RR to 35. • If pH remains < 7.15, VT may be increased in 1 ml/kg steps until pH > 7.15 (Pplat target of 30 may be exceeded). • May give NaHCO3 • Alkalosis Management: (pH > 7.45) Decrease vent rate if possible.
- 36. When to wean?
- 37. Weaning Conduct a SPONTANEOUS BREATHING TRIAL daily when: 1. FiO2 ≤ 0.40 and PEEP ≤ 8 OR FiO2 < 0.50 and PEEP < 5. 2. PEEP and FiO2 ≤ values of previous day. 3. Patient has acceptable spontaneous breathing efforts. (May decrease vent rate by 50% for 5 minutes to detect effort.) 4. Systolic BP ≥ 90 mmHg without vasopressor support. 5. No neuromuscular blocking agents or blockade.
- 38. Weaning proper If all above criteria are met and subject has been in the study for at least 12 hours, initiate a trial of UP TO 120 minutes of spontaneous breathing with FiO2 < 0.5 and PEEP < 5: 1. Place on T-piece, trach collar, or CPAP ≤ 5 cm H2O with PS < 5 2. Assess for tolerance as below for up to two hours. a. SpO2 ≥ 90: and/or PaO2 ≥ 60 mmHg b. Spontaneous VT ≥ 4 ml/kg PBW c. RR ≤ 35/min d. pH ≥ 7.3 e. No respiratory distress (distress= 2 or more of the following)
- 39. Weaning proper Feature of respiratory distress: a) HR > 120% of baseline b) Marked accessory muscle use c) Abdominal paradox d) Diaphoresis e) Marked dyspnea 3. If tolerated for at least 30 minutes, consider extubation. 4. If not tolerated resume pre-weaning settings. 5.?? NIV
- 40. Other approaches
- 41. Other approaches • Prone Positioning: Reduces ventral to dorsal trans pulmonary pressure difference. Improves recruitment of dependent lung zones And also improves VP mismatch. • Should be done for > 12 hrs a day • Inverse ratio ventilation- I:E ratio (Upto 7:1) and lower peak pressures • High frequency Oscillatory Ventilation: Low Tidal Volumes (1-2 ml/Kg) are given at higher RR. These reduce distention and prevent VALI. • Airway Pressure Release Ventilation: Long Duration-High Pressure followed by Short Duration and Lower Pressure.
- 42. Other approaches • ECMO • Recruitment maneuvers that transiently increase PEEP to high levels to “recruit” atelectatic lung can increase oxygenation.
- 43. Fluid Management-2 Approaches • Aggressive attempts to reduce left atrial filling pressures with fluid restriction and diuretics should be an important aspect of ARDS management, limited only by hypotension and hypoperfusion of critical organs such as the kidneys. • Non pulmonary cause of ARDS on the other hand usually benefit from a fluid liberal approach.
- 44. Other pharmacotherapies 1. NEUROMUSCULAR BLOCKADE • In severe ARDS, sedation alone can be inadequate for the patient- ventilator synchrony required for lung-protective ventilation. • Early administration of neuromuscular blockade if needed to facilitate mechanical ventilation in severe ARDS improves survival and reduces ICU stay. 2. Inhaled pulmonary vasodialators (NO, inhaled prostacyclins- epoprostenol) • Increase oxygenation • No mortality benefit • Not a part of guidlines
- 45. Other pharmacotherapies 3. Antibiotics ? Benefit -Beta 2 agonists -Sivelestat: Neutrophil elastase inhibitor -Simvastatin
- 46. Glucocorticoids • Anti inflammatory action • Recent evidence has shown improved mortality, better outcomes in patients with moderate and severe ARDS • Usual doses: • Moderate ARDS-0.5 to 1 mg/kg for 5 to 10 days • Severe ARDS-1 to 2 mg/kg for 5 to 10 days • Yet to be formally incorporated in guidelines
- 47. Prognostic Markers 1. Severity as per Berlin definition 2. Delta P = P Plateau - PEEP
- 48. Thank You