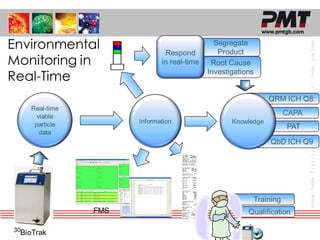
The document discusses proposed changes to Annex 1 regarding viable monitoring of cleanrooms. It notes that Annex 1 will now require continuous monitoring throughout critical processes, rather than interval sampling. This reflects a shift from air sampling to real-time air monitoring. Continuous active air monitors are presented as able to meet the new Annex 1 requirements, capturing transient events better than settle plates. The document discusses various active air monitoring devices and their particle collection capabilities compared to traditional samplers. Real-time microbial monitoring using laser induced fluorescence is also proposed as an alternative to growth-based methods.