This document discusses environmental microbial monitoring (EMM) in cleanrooms and pharmaceutical facilities. It provides an overview of EMM purposes and regulations, who performs EMM, what areas are monitored, sampling plans and methods used. Key points covered include:
- EMM determines microbial and particulate levels to ensure cleanroom quality and identify contamination sources.
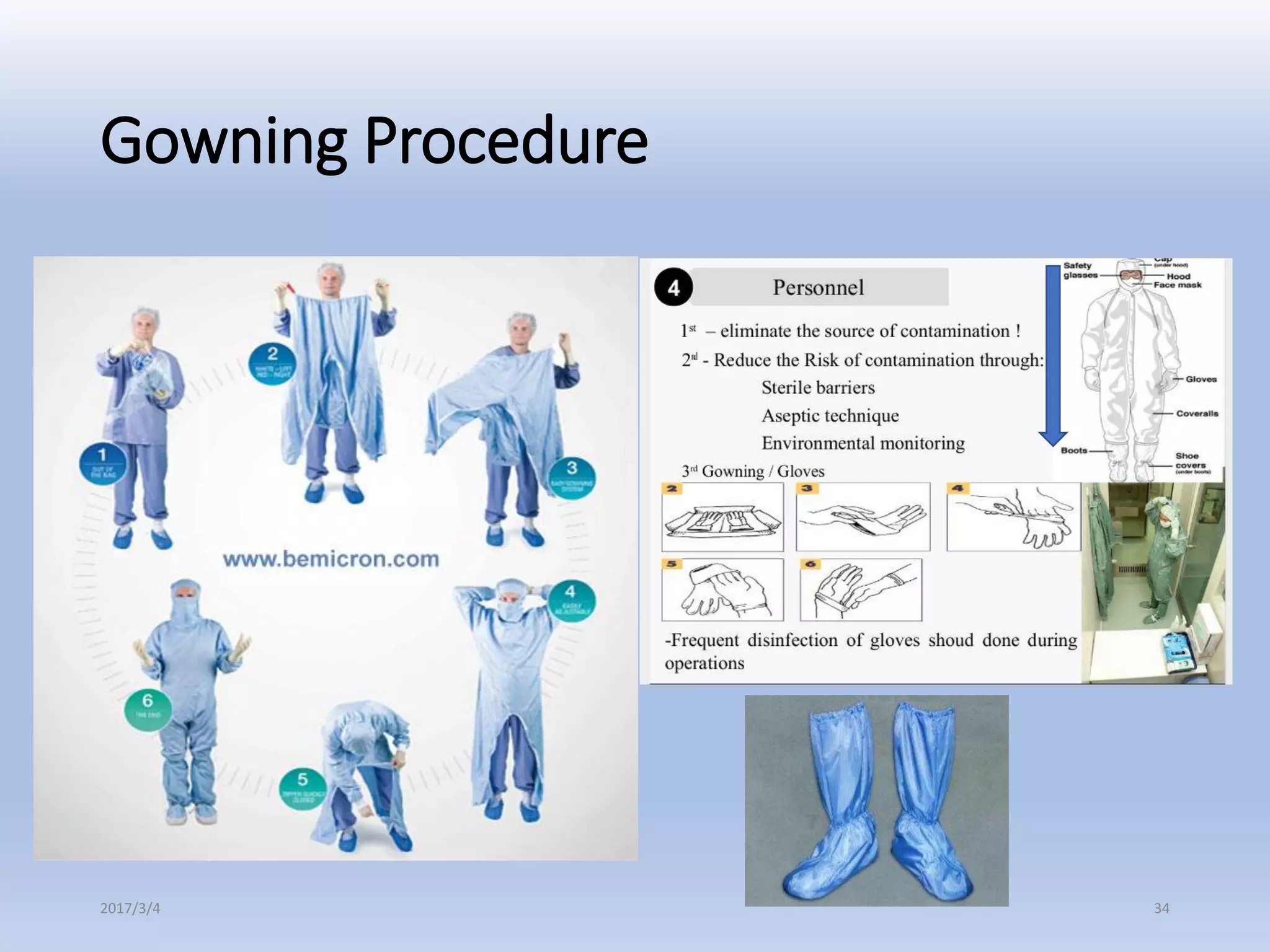
- Quality Control and Assurance departments perform EMM to demonstrate safety and ensure GMP compliance.
- Non-viable air, viable air and surface samples are monitored from areas like personnel, equipment and facilities.
- Sampling frequency, sites and methods like air samplers, settle plates, contact plates and swabbing are discussed in accordance with regulations like USP 39