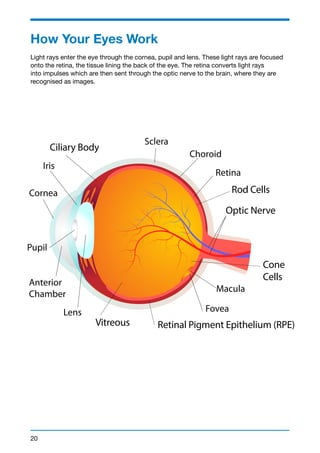
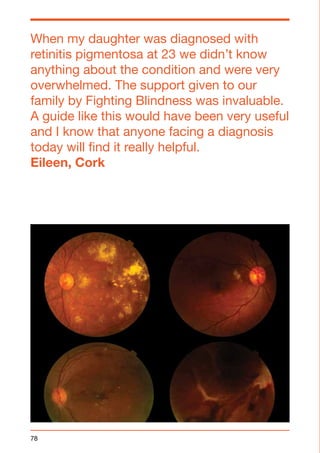
This document provides a summary of conditions that affect the retina. It begins with an introduction from the CEO of Fighting Blindness, noting that over 224,000 people in Ireland are affected by sight loss. It then discusses Fighting Blindness's mission to cure blindness, support those with sight loss, and empower patients. The document provides information on various retinal conditions, including age-related macular degeneration, diabetes-related sight loss, rare inherited conditions like retinitis pigmentosa and Stargardt disease, and Fighting Blindness' research and support services. It aims to be a valuable resource for individuals and families affected by retinal conditions.