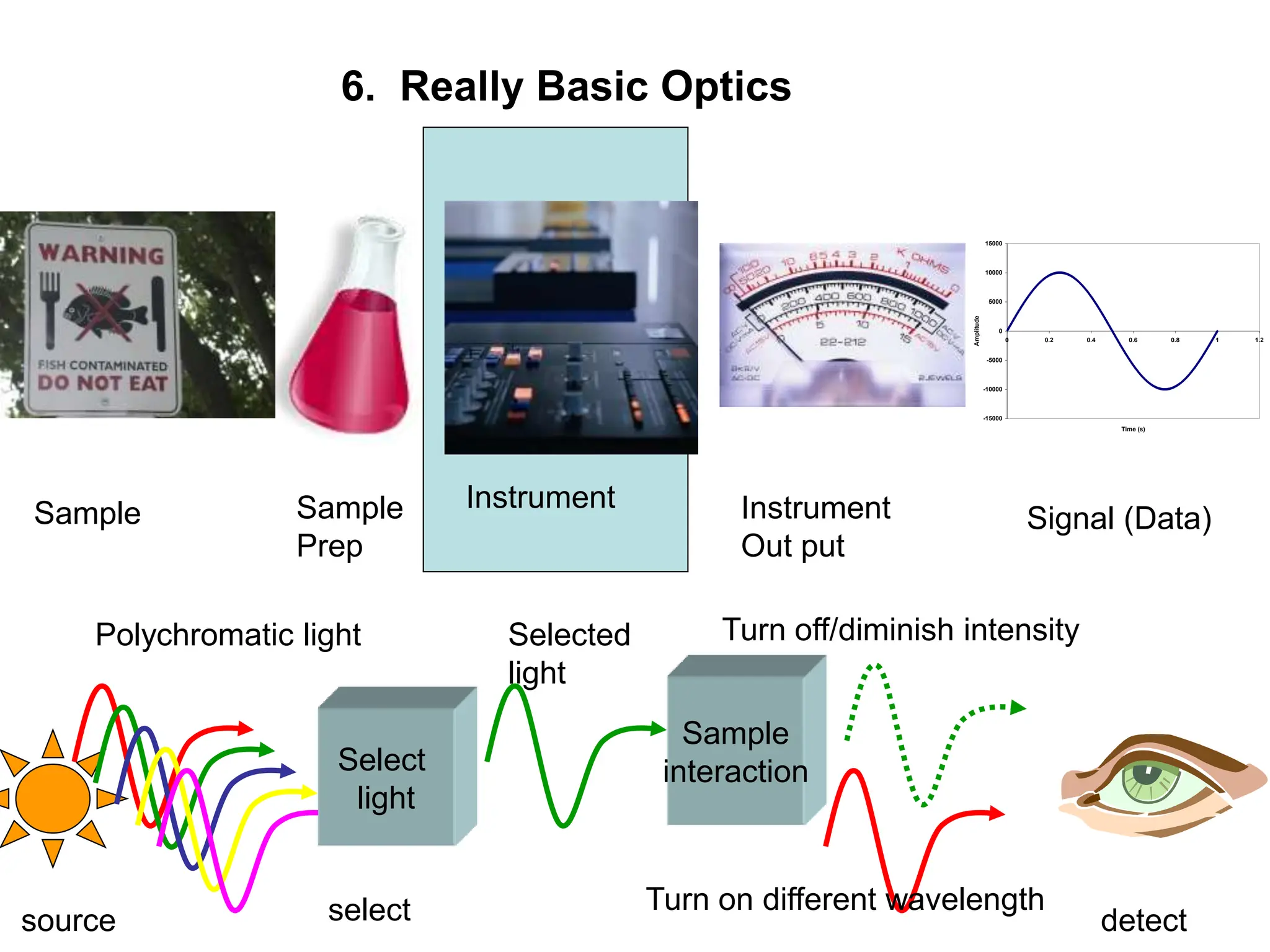
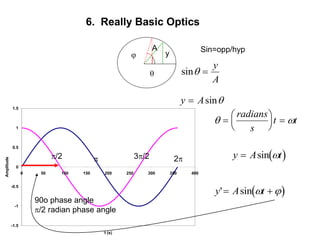
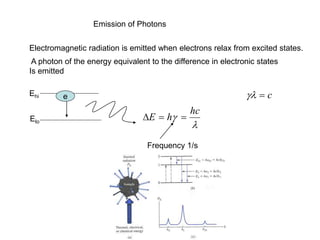
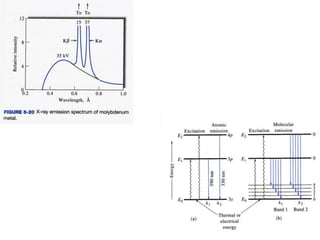
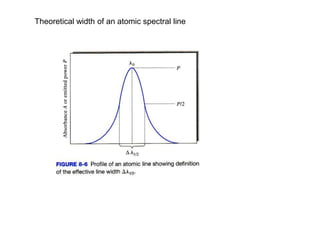
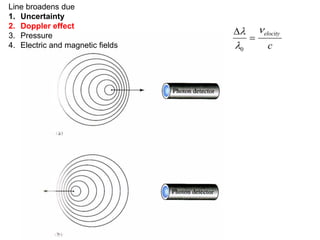
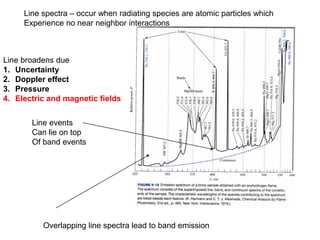
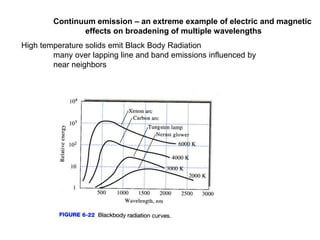
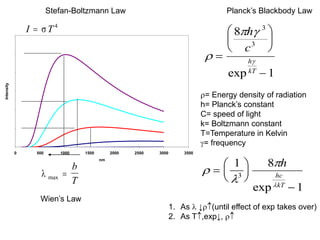
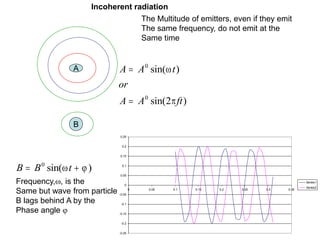
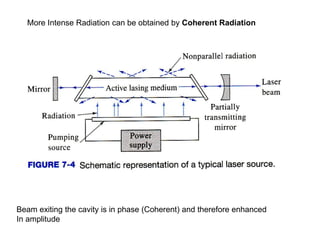
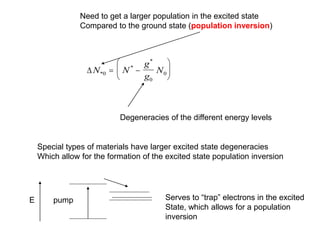
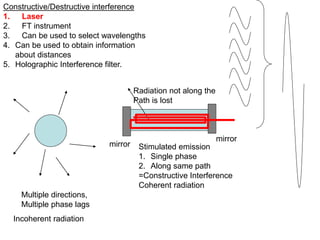
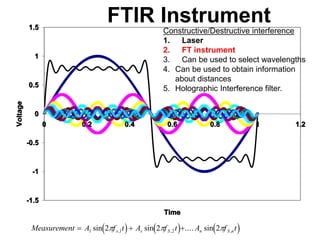
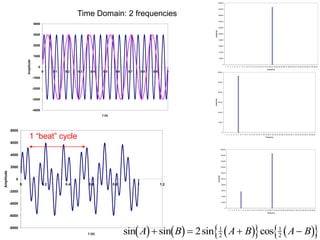
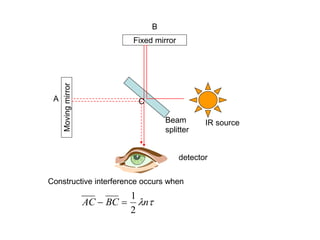
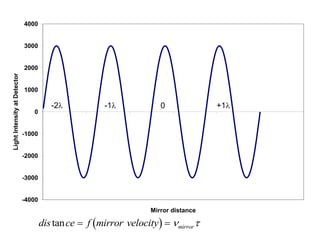
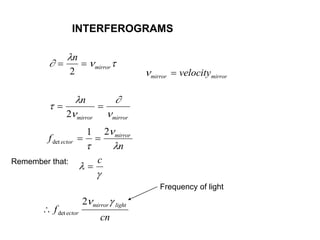
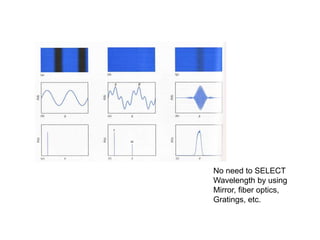
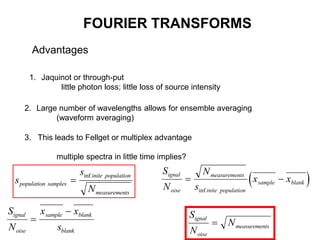
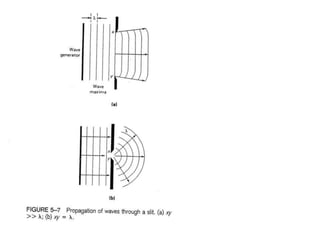
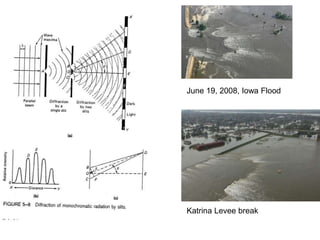
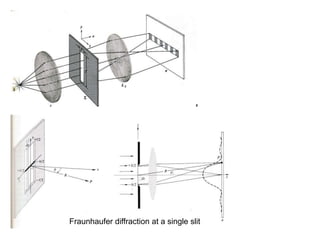
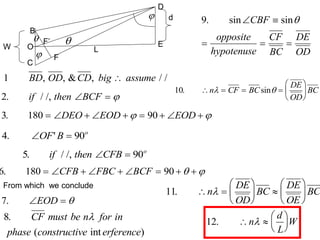
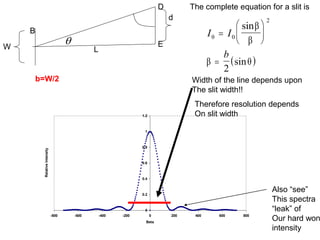
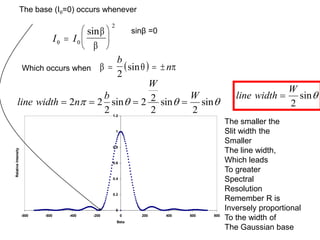
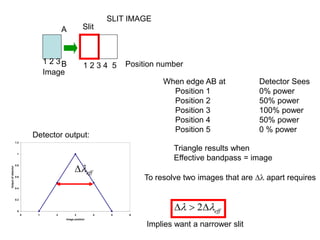
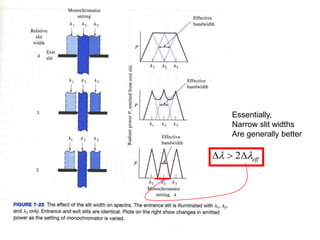
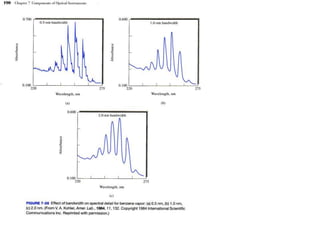
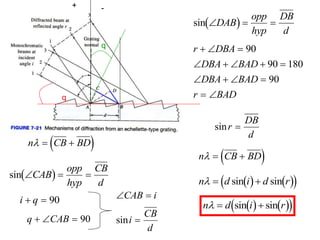
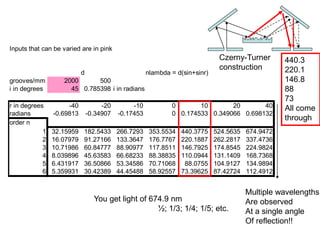
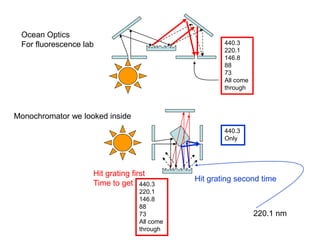
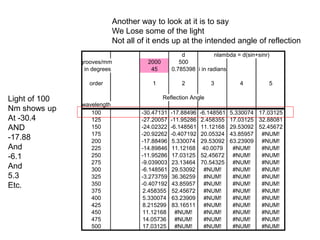
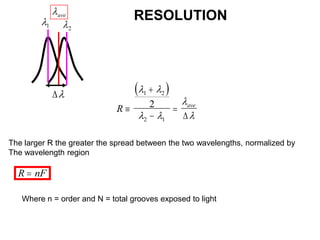
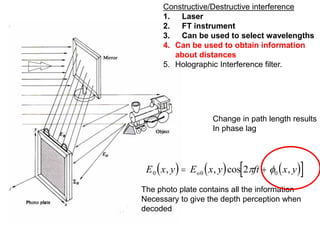
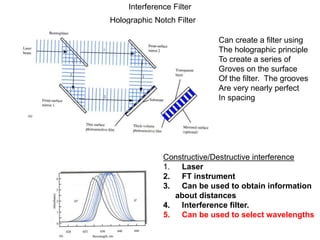
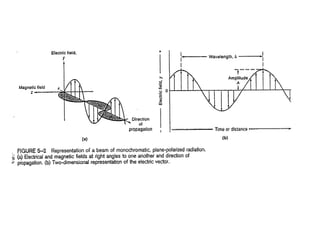
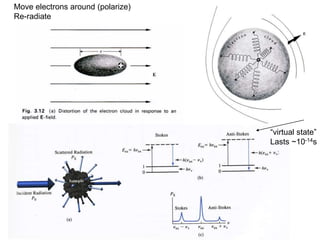
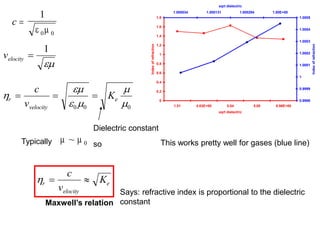
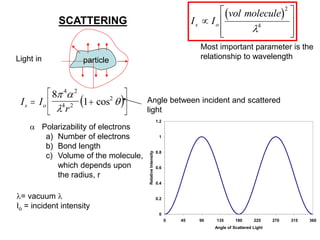
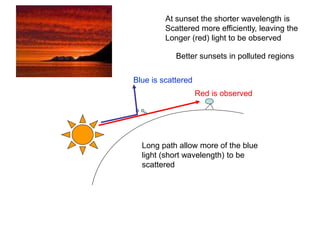
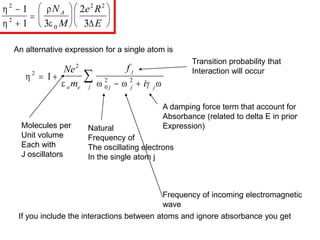
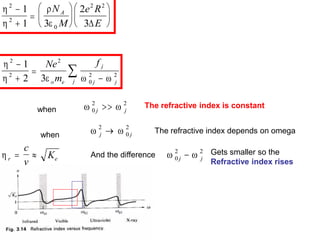
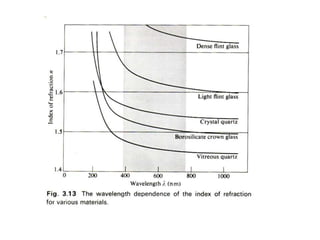
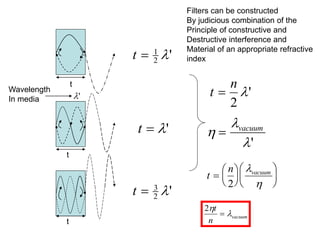
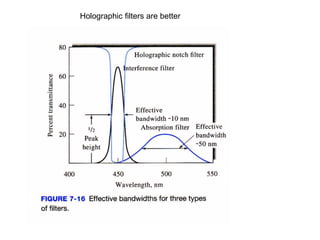
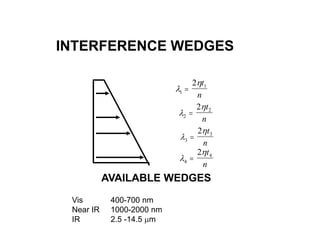
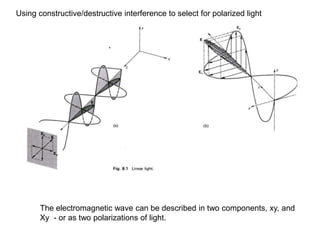
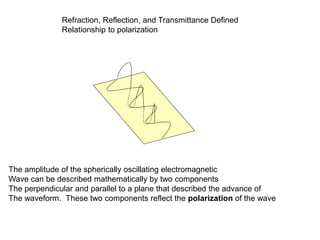
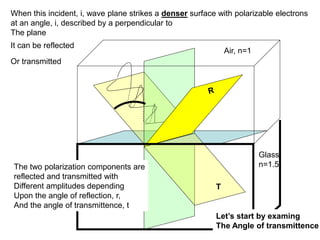
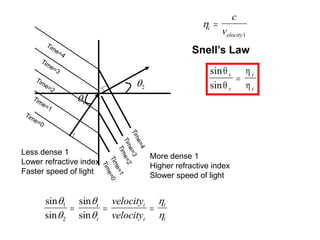
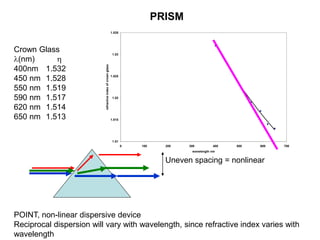
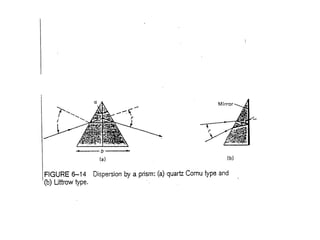
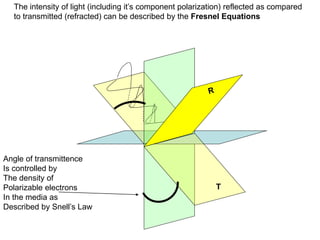
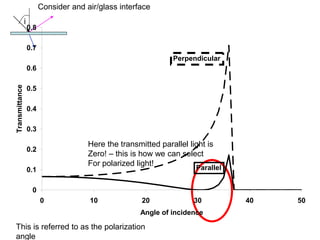
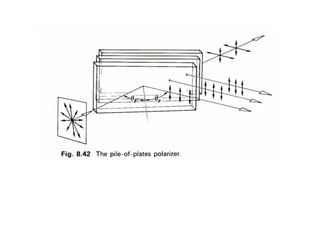
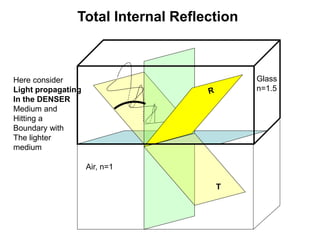
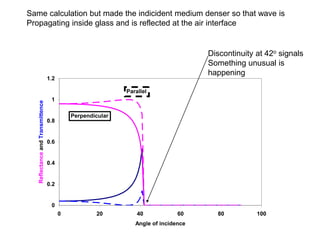
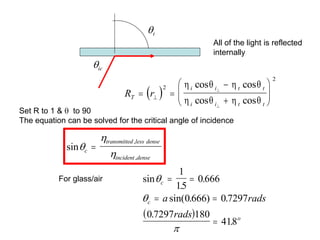
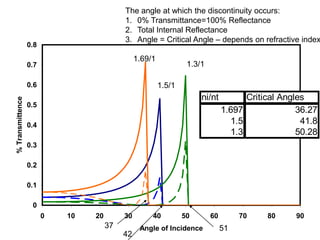
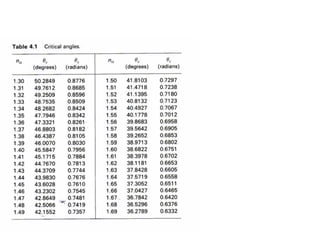
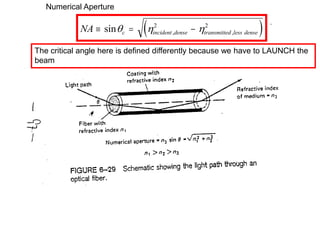
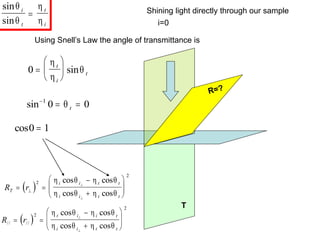
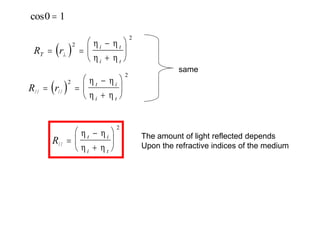
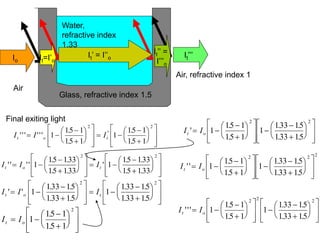
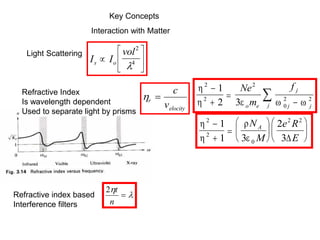
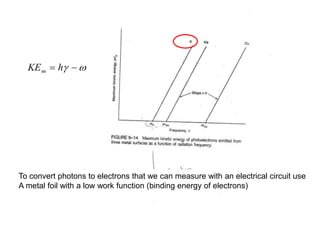
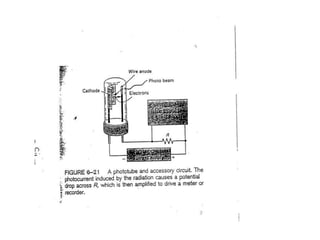
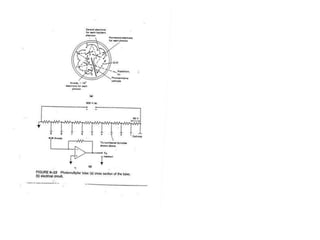
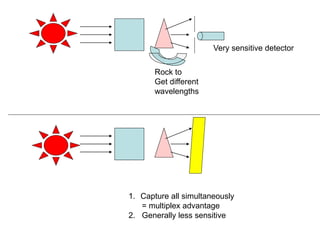
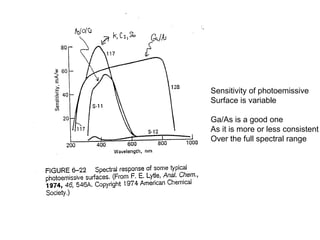
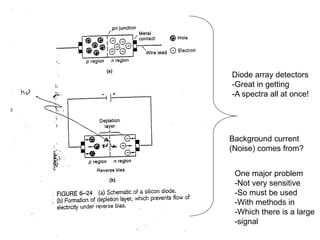
This document provides an overview of really basic optics concepts. It defines key terms like phase angle and discusses topics like atomic line widths, Doppler broadening, molecular bands, and continuum sources. It also covers the differences between coherent and incoherent radiation, how constructive and destructive interference can be used in lasers and Fourier transform instruments, and how population inversion allows for light amplification in lasers.