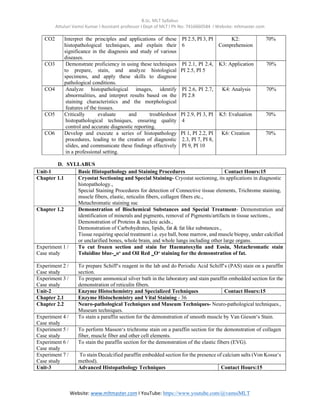
The document outlines the syllabus for the B.Sc. in Medical Lab Technology, focusing on the course 'Applied Histopathology - II', which builds on foundational histopathological skills by teaching various staining procedures and techniques. It details course objectives, outcomes, and specific units covering basic histopathology, enzyme histochemistry, and advanced techniques, each supported by practical experiments. The document also lists performance indicators and reference materials for students and educators in the field.