12. meristem culture
- 1. MERISTEM CULUTRE PTC course for Horticulture students. By: Dr. Rafail S. Toma 1
- 2. What is meristem? A meristemis the tissue in all plants consisting of undifferentiated cells (meristematic cells), found in zones of the plant where growth can take place. PTC course for Horticulture students. By: Dr. Rafail S. Toma 2
- 3. What is meristem? Groups of cells that are the source of new cells form tissue called meristem. Meristemcells aren't specialized, but when they divide, some of the new cells specialize into tissues. Areas of growth that lengthen the tips of roots and stems are called apical meristems. Lateral meristems, found all along woody roots and stems, increase the thickness of these plant parts. PTC course for Horticulture students. By: Dr. Rafail S. Toma 3
- 4. Meristem Culture History Though meristemculture technique is known since 1933 it was made successful only in 1965 by Morel. In the early period (1949) of this adventure, Wetmore and Morel regenerated plantlets fromthe meristemof ferns on simple defined medium. But this did not workwell with the angiosperms since they required complex mediumfortheirin vitro development. PTC course for Horticulture students. By: Dr. Rafail S. Toma 4
- 5. Meristem Culture History In 1946, Ball found the initiation of root primordiumformation and the plantlets regeneration fromthe young meristems of Trpaeolummajus and Lupinus albus. Following this, Morel established the technique of meristemculture with orchids. Of late meristemculture technique is being increasingly applied in micropropagation as an alternative means forsexual propagation of economically important crop plants. PTC course for Horticulture students. By: Dr. Rafail S. Toma 5
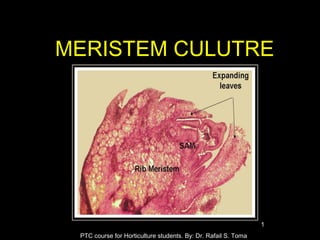
- 6. Explants The explant of meristemculture may eitherbe the apical dome (apical meristem) ormore frequently, the apical dome plus a few subjacent leaf primordia (the sub apical meristematic region). The apical meristemis located at the extreme tip of a shoot and measure 0.1 mmin diameterand 0.25 to 0.30 in length. PTC course for Horticulture students. By: Dr. Rafail S. Toma 6
- 7. The culture of meristeminvolves three stages: 1. culture establishment, 2. multiplication of the propagules and 3. root regeneration PTC course for Horticulture students. By: Dr. Rafail S. Toma 7 MeristemCulture Technique
- 8. Culture can be established from meristem, shoot tips or axillary buds. Forshoot regeneration from meristem, young development stage of meristemhas been found to be optimum. Therefore, it is desirable to excise terminal explants for culture. Axillary buds are preferred since there would be only one terminal bud pershoot. Further, the explants should be largerenough forgetting successful results. So largerexplants like shoot tips and buds have to be chosen instead of minute meristems. PTC course for Horticulture students. By: Dr. Rafail S. Toma 8 Culture Establishment
- 9. Afterexplant excision, they are inoculated into culture medium. Generally there is no necessity forthe addition of exogenous hormones in the mediumsince sufficient quantity of endogenous hormone is present in the shoot apices. However, there are cases in which exogenous auxin is applied to get betterresults. Among the auxins, NAA is the auxin routinely used for meristemtip and bud cultures. PTC course for Horticulture students. By: Dr. Rafail S. Toma 9 Culture Establishment
- 10. The propagules multiplication in meristem/shoot tip/axillary bud culture can be accomplished by the three methods as given below: Explant - callus - meristemoids -shoot/roots plantlets Explant - callus - embryoids/embryos- plantlets Explant – axillary buds - multiple shoots-roots plantlets PTC course for Horticulture students. By: Dr. Rafail S. Toma 10 Multiplication of propagules
- 11. Among the three methods, axillary shoot proliferation is considered as the best because of the lowerriskof genetic instability than the other two systems of multiplication and is easily achievable in most plant species. In this system, the concentration of cytokinin used is comparatively higherand is done to overcome the apical dominance. PTC course for Horticulture students. By: Dr. Rafail S. Toma 11 Multiplication of propagules
- 12. The incorporation of cytokinin enhances the branching of lateral buds from leaf axils. Too high a concentration of auxin may not only inhibit axillary bud branching but also induces callus formation, especially when 2,4-Dis used. PTC course for Horticulture students. By: Dr. Rafail S. Toma 12 Multiplication of propagules
- 13. The purpose of this stage, in the meristemculture is to induce regeneration of roots fromthe shoot multiplied in the previous stage. Adventitious root formation can be induced quite readily in many species, but it can be very much recalcitrant in most woody species. PTC course for Horticulture students. By: Dr. Rafail S. Toma 13 Roots Regeneration
- 14. The induction of rooting does not always have to be carried out invitro. Good rooting can be obtained in greenhouse by placing shoots into pasteurized sand underintermittent mist. Forbetterrooting, the proliferated shoots may be dipped in auxin solution orcommercial rooting powderbefore planting into rooting medium. PTC course for Horticulture students. By: Dr. Rafail S. Toma 14 Roots Regeneration
- 15. Size of the explant The size of the explant determines the survival of the culture. In general, the largerthe explant, the betterthe chance of survival. Meristemof the smallest size within the regenerable range should be used forvirus elimination. When very small explants are used, the presence of leaf primordia appears to determine the capability of an explant to develop. PTC course for Horticulture students. By: Dr. Rafail S. Toma 15 Factors influencing meristem culture
- 16. Physiological state of the explant Explants taken fromthe tip of a shoot are in a younger stage of development than explants taken fromthe base. Young developmental stage has often been found to be optimumforbettershoot regeneration. PTC course for Horticulture students. By: Dr. Rafail S. Toma 16 Factors influencing meristem culture
- 17. Culture media White's medium(1943) was the most widely used medium during the early days of meristemculture. There is no general purpose mediumyet available for meristem, shoot tip and bud culture. Murashige and Skoog (1962) mediumwith some modification is the one used more frequently and with great success. PTC course for Horticulture students. By: Dr. Rafail S. Toma 17 Factors influencing meristem culture
- 18. Growth regulators The requirement of growth regulators varies form species to species, fromone stage of culture development to another. Presence of cytokinin at higherlevel during proliferation stage is felt to overcome the apical dominance. Similarly, presence of auxin is headed forgood rooting. PTC course for Horticulture students. By: Dr. Rafail S. Toma 18 Factors influencing meristem culture
- 19. A. Invitro micropropagation, B. Production of pathogen free plants, and C. Cryopreservation of germplasm. PTC course for Horticulture students. By: Dr. Rafail S. Toma 19 Applications of meristem culture
- 20. A. Invitro micropropagation The micropropagation technique through meristem orshoot tip culture favors production of thousand and thousands of plants froma single explant within a short period. Moreover, once a stockof multiple shoot culture is established, it can continuously serve as the source material instead of having to restart fromfresh explant cultures periodically. PTC course for Horticulture students. By: Dr. Rafail S. Toma 20 Applications of meristem culture
- 21. A. Invitro micropropagation The greatest success using this technique has been achieved in most of the herbaceous horticultural species. Compared to herbaceous plants, the micropropagation of woody species has lagged far behind. PTC course for Horticulture students. By: Dr. Rafail S. Toma 21 Applications of meristem culture
- 22. A. Invitro micropropagation The majorproblems encountered with the propagation of woody species are: 1. Most of the forest species are recalcitrant to culture condition because of the presence of large quantity of polyphenolic compounds in the tissues and 2. The otherdifficulty experienced is rooting of in vitro cultures. PTC course for Horticulture students. By: Dr. Rafail S. Toma 22 Applications of meristem culture
- 23. B. Production of pathogen free plants The most important application of micropropagation technique viameristemculture is the production of pathogen free plants, especially viruses as they are absent in apical meristem. PTC course for Horticulture students. By: Dr. Rafail S. Toma 23 Applications of meristem culture
- 24. B. Production of pathogen free plants Generally, viruses infect plant species systemically making the plants to die. But the evidences fordecrease in virus particles toward apical meristemmade Morel and Martin (1952) to postulate the concept of culturing apical meristemof systemically infected plant invitro in orderto obtain virus free plants, genetically identical to the "motherplant” PTC course for Horticulture students. By: Dr. Rafail S. Toma 24 Applications of meristem culture
- 25. B. Production of pathogen free plants Forexample potato virus Xinfection could not be totally eradicated since these viruses maintain theirreplication in actively growing meristem. In some cases of meristemtip culture the heat therapy has necessarily to be followed to eliminate the viruses. Forexample, in the carnation, heat therapy of plants at 38°C fortwo months followed by meristem culture eradicated all the viruses. PTC course for Horticulture students. By: Dr. Rafail S. Toma 25 Applications of meristem culture
- 26. B. Production of pathogen free plants The proposed reasons formeristem’s virus freeness: 1. The absence of vascularconnections 2. The high metabolic activity of the meristematic cells which prevent virus multiplication 3. The high activity of the affective virus abolishing group in meristems 4. The high auxin levels in apical meristems inhibits virus multiplication PTC course for Horticulture students. By: Dr. Rafail S. Toma 26 Applications of meristem culture
- 27. C. Cryopreservation of germplasm The conventional systemof seed storage has the following disadvantages: 1) the loss of viability of seeds, 2) destruction by pathogen and pest attacks, 3) problems in clonally propagated crops, 4) high cost of maintenance and transport and 5) material loss due to environmental hazards. PTC course for Horticulture students. By: Dr. Rafail S. Toma 27 Applications of meristem culture
- 28. C. Cryopreservation of germplasm Considering the above disadvantages, the feasibility of invitro storage was extensively studied. The potential advantages of this method are: 1) relatively little space is needed, 2) the plants are maintained free frompest and pathogens, PTC course for Horticulture students. By: Dr. Rafail S. Toma 28 Applications of meristem culture
- 29. C. Cryopreservation of germplasm 3) maintenance of vegetatively propagated species is easier, 4) the materials can be multiplied as and when needed and 5) the pest pathogen free nature favors easy and quickinternational germplasmexchanges. PTC course for Horticulture students. By: Dr. Rafail S. Toma 29 Applications of meristem culture
