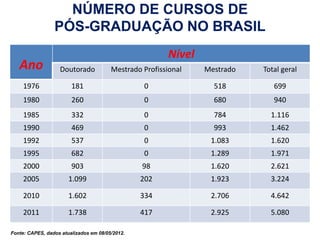
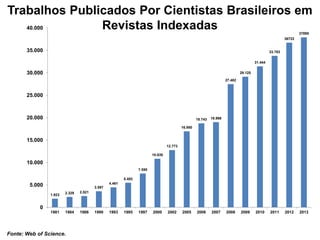
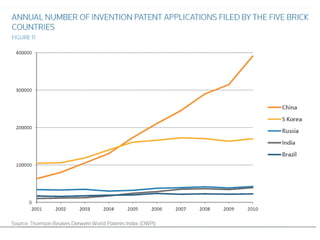
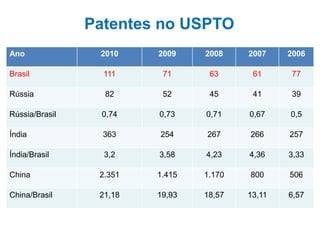
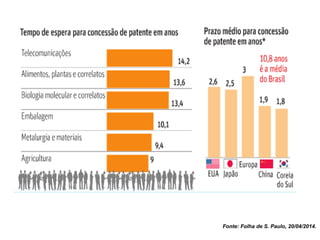
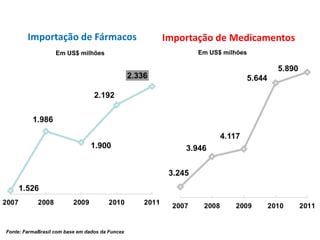
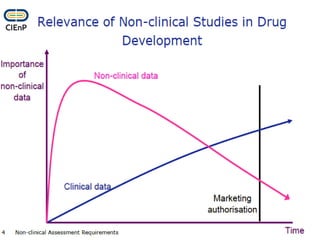
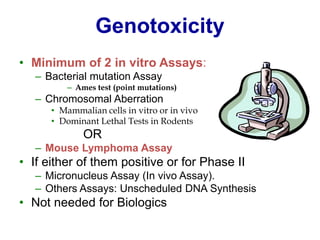
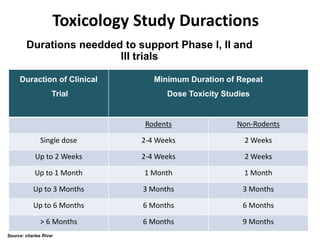
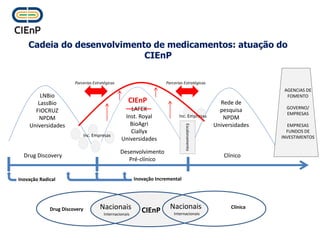
This document discusses innovation and pre-clinical development in Brazil. It outlines characteristics of innovative countries, recent advances in science and technology in Brazil, and the growth of post-graduate programs. It then discusses the importance of pre-clinical studies like toxicology, pharmacokinetics, safety pharmacology, and local tolerance to support clinical trials. Finally, it introduces the Centre of Innovation and Pre-Clinical Studies, which aims to conduct pre-clinical research, support pharmaceutical development, and contribute to generating national competence in drug innovation.