Inhibition of enzyme action
- 2. Introduction • Enzyme, a protein molecule, acts as a catalyst in a reaction. • Enzyme inhibition- A science of enzyme substrate reaction influenced by the presence of any organic chemical, inorganic metal or biosynthetic compound due to their covalent or non-covalent interactions with the enzyme active site. • Enzyme inhibitors- low molecular weight compounds that bind to an enzyme and decrease its activity.
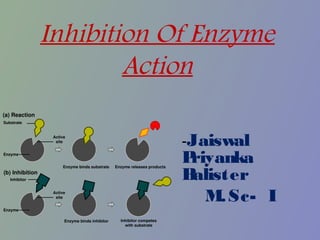
- 3. • Enzyme inhibitors can be specific or non-specific. • Specific inhibitors can reduce or completely inhibit the enzyme catalytic activity either reversibly or irreversibly (permanently). • Reversible inhibition can be: - Competitive - Uncompetitive - Non-competitive • Poisons [such as cyanide, carbon monoxide and polychlorinated biphenols (PCBs)] and drugs are examples of enzyme inhibitors.
- 4. Competitive Inhibitors • A competitive inhibitor is any compound which closely resembles the chemical structure and molecular geometry of the substrate. • The inhibitor competes for the same active site as the substrate molecule. • The inhibitor may interact with the enzyme at the active site, but no reaction takes place. • Because of the presence of the inhibitor, fewer active sites are available to act on the substrate.
- 5. • Since the enzyme's overall structure is unaffected by the inhibitor, it is still able to catalyze the reaction on substrate molecules that do bind to an active site. • Since the inhibitor and substrate bind at the same site, competitive inhibition can be overcome simply by raising the substrate concentration. • Therefore, the amount of enzyme inhibition depends upon the inhibitor concentration, substrate concentration, and the relative affinities of the inhibitor and substrate for the active site.
- 6. Reaction Scheme • The reaction scheme that corresponds to competitive inhibition is: • The inhibitor reduces the amount of E available for productive catalysis by the formation of the EI complex. The inhibitor does not affect the ES complex after it has formed. The dissociation constant for the inhibitor is KI = [E][I]/[EI].
- 7. Michaelis-Menten curve & Lineweaver Burk Plot The Lineweaver-Burk plot shows an unchanged Vmax and a slope that increases with inhibitor concentration.
- 8. • The Lineweaver-Burk plot shows an unchanged Vmax and a slope that increases with inhibitor concentration. • Steady-state analysis of the effect of the inhibitor shows that KM is increased by a factor of (1 + [I]/KI). The resulting form of the Michaelis- Menten equation is:
- 9. Disulfiram (Antabuse) • The drug inhibits the aldehyde oxidase which causes the accumulation of acetaldehyde with subsequent unpleasant side-effects of nausea and vomiting. • This drug is sometimes used to help people overcome the drinking habit.
- 10. Ethanol • Methanol poisoning occurs because the enzyme alcohol dehydrogenase oxidizes it to formaldehyde, CH2O, which is a potent poison and formic acid which attack the optic nerve causing blindness. • Ethanol is given as an antidote for methanol poisoning because ethanol is a competitive inhibitor of methanol to alcohol dehydrogenase. • Ethanol is oxidized in preference to methanol and consequently, the oxidation of methanol is slowed down so that the toxic by-products do not have a chance to accumulate.
- 11. References 1. CHEMICAL KINETICS, 3rd Ed., Keith J. Laidler, Pearson Publication. 2. BIOCHEMISTRY, 5th Ed., Richard Harvey & Denise Ferrier. 3. PHYSICAL CHEMISTRY FOR THE BIOSCIENCES, Raymond Chang. 4. PRINCIPLES AND TECHNIQUES OF PRACTICSL BIOCHEMISTRY, 5th Ed., Keith Wilson & John Walker.
