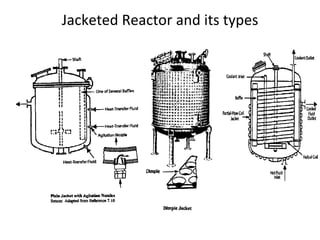
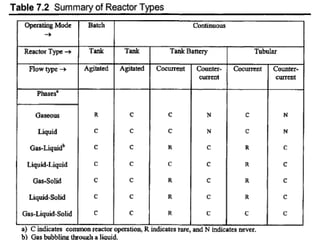
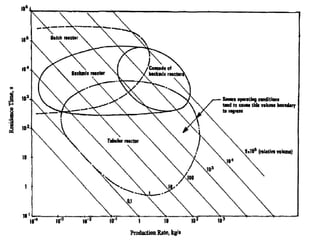
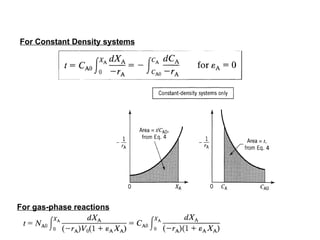
The document discusses various topics related to chemical reactor design including:
1. Reactor classification into homogeneous and heterogeneous types and examples like batch, continuous stirred tank, plug flow, and semi-batch reactors.
2. Factors to consider for reactor design like heat of reaction, operating temperature and pressure, and use of internal or external heating/cooling.
3. Methods for controlling temperature like adiabatic, isothermal, auto-thermal reactors.
4. Key principles of chemical equilibrium and kinetics that influence choice of process conditions.