Chemistry topic Prions
- 1. Government college University of Faisalabad Department of bio- informatics By: Khansa Asad Prions
- 2. Introduction: The term PRION is derived from proteinacioues infectious particle and refer to the pathogen that cause "Transmissible spongiform encephalopathies"(TsFs). A prion is a type of protein that can trigger normal proteins in the brain to fold abnormally. Prion diseases can affect both humans and animals and are sometimes spread to humans by infected meat products. The most common form of prion disease that affects humans is Creutzfeldt-Jakob disease (CJD) Discovery: PRION is a infectious disease. Prion recently discovered by American Neurobiologist"STANLEY Prusiner". Prion is the least understood Micro organism Nature: Nature of PRION is CONTROVERSIAL. Composed of: Prion is composed of protein only that contains the information and codes for their own replication. Responsibility: responsible for the mad cow infection. for the my mysterious brain infection in man Function: The physiological function of PRION protein remains a CoNTROVERSIAL matter. While data from in vitro experiment suggests may dissimilar roles,studies on PrP knockout mice have provided only limited information because these Animals exhibit only minor abnormalities.The research done in mice it was found that the cleavage of PrP protein in peripheral nerves causes the Activation of myelin repair in Sechwan Cells and that the Lack of PrP protein caused Demyelination in these cells. Prions are misfolded proteins with the ability to transmit their misfolded shape onto normal variants of the same protein. They characterize several fatal and transmissible neurodegenerative diseases in humans and many other animals.
- 3. Prion replication mechanisms: The firsthypothesisthattriedtoexplainhow prionsreplicate inaproteinonlymannerwasthe HeterodimerModel. Thismodel assumedthata single PrPscmolecule bindstoasingle PrPcmolecule andCatalyzesit's conversion intoPrPsc.The twoPrPscmoleculethencome apartandcan go onto convertmore PrPc.
- 4. Manfredeigenshowthatthe heterodimermodel requiresPrPsctobe an extraordinaryeffective Catalyzes,increasingthe rate of conversionreactionbyafactor of around 10(15). The mechanismsof PRION replicationhasimplicationsof designingdrugs.
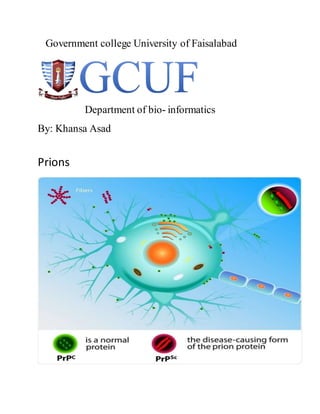
- 5. Structure: The protein that prions are made Of PRION protein(prp) is found Throughout the body,even in healthy People and in animals. However,PrP found in infectious material has a Different structures and is resistance to Proteases ,the enzymes in the body that can normallyBreakdown protein.The Normal from of protein is called PrPc.While infectious from is called PrPsc. The' C' refers to cellular and while the'Sc'Refers to" scrapie" Won prize: Prusiner won the" Nobel prize" of Physiology or medicine in 1997 for his researchers into prions. PRION diseases and their transmission: AffectedAnimals(s): Diseases: Sheepor Goat:- causes the: "SCRAPIE" Cattle:- Bovine spongiformencephalopathies (BSE)-"madcow" diseases Mink:- Transmissible mink,encephalopathy (TME). White-taileddeer,mule,elk, Deer,moose:- ChrONICWASTING Diseases(CWD). Cat:- Feline spongiformencephalopathy (FSE).
- 6. Nyala,OryX, Greaterkudu:- These animals are also effected By prions as Exoticungulated Encephalopathy(EUE). Ostrich:- Spongiformencephalopathy (Has not beenshown to be transmissible) Humans:- -Creutzfeld-jakobdiseases(CJD). -variant creutzfeld-jakobdisease(VcJD) -latrogeniccreutzfeld-jakobdisease(ICJD). -familial creutzfeld-jakobdisease(fcJD). -sporadic-straussler-schienkersyndrome(Gss). -fatel familial insomnia(FFI). -Familial spongiformencephalopathy -Multiple systemAtrophy(MSA) Not a TSE and isnot by typical prions PrP/PrPsc,but by a misfoldedas Alpha-synuclein. PrP and long-term memory:- A review of the evidence in 2005 suggests ,That PrP may have a normal function In maintenance of "LonG-Term Memory" As well a,2004 study found thatMice lacking genes for normal Cellular PrP protein show altered Hippocampal lonG-term potentiation.
- 7. Types:- 1-PrPc 2-PrPsc The Normal from of protein is called the PrPc. While the infectious protein from is called the PrPsc.