Periodic table by Cassidy Estabrooke
•Download as KEY, PDF•
1 like•620 views
Report
Share
Report
Share
More Related Content
What's hot
What's hot (20)
Ch 5.1,5.2 organizing elements & the periodic table

Ch 5.1,5.2 organizing elements & the periodic table
The periodic table presentation for 4050 [autosaved]![The periodic table presentation for 4050 [autosaved]](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![The periodic table presentation for 4050 [autosaved]](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
The periodic table presentation for 4050 [autosaved]
Short Metal-Metal Bond Distances Containing the Indium tris(3,5-dimethyl)-1 P...

Short Metal-Metal Bond Distances Containing the Indium tris(3,5-dimethyl)-1 P...
Similar to Periodic table by Cassidy Estabrooke
Similar to Periodic table by Cassidy Estabrooke (20)
Class 10. Chapter 5. Periodic Classification of Elements

Class 10. Chapter 5. Periodic Classification of Elements
01-VI_OLY_CHE_PERODIC CLASSIFICATION_VOL-4_PG-1 TO 45.pdf

01-VI_OLY_CHE_PERODIC CLASSIFICATION_VOL-4_PG-1 TO 45.pdf
Презентация по химии _Д.И.Менделеев и его великое открытие_ (на английском яз...

Презентация по химии _Д.И.Менделеев и его великое открытие_ (на английском яз...
Periodic calssification of elements ncert shashikumar b s

Periodic calssification of elements ncert shashikumar b s
Recently uploaded
APM Welcome, APM North West Network Conference, Synergies Across Sectors

APM Welcome, APM North West Network Conference, Synergies Across SectorsAssociation for Project Management
Recently uploaded (20)
Measures of Central Tendency: Mean, Median and Mode

Measures of Central Tendency: Mean, Median and Mode
social pharmacy d-pharm 1st year by Pragati K. Mahajan

social pharmacy d-pharm 1st year by Pragati K. Mahajan
Call Girls in Dwarka Mor Delhi Contact Us 9654467111

Call Girls in Dwarka Mor Delhi Contact Us 9654467111
Beyond the EU: DORA and NIS 2 Directive's Global Impact

Beyond the EU: DORA and NIS 2 Directive's Global Impact
Disha NEET Physics Guide for classes 11 and 12.pdf

Disha NEET Physics Guide for classes 11 and 12.pdf
APM Welcome, APM North West Network Conference, Synergies Across Sectors

APM Welcome, APM North West Network Conference, Synergies Across Sectors
JAPAN: ORGANISATION OF PMDA, PHARMACEUTICAL LAWS & REGULATIONS, TYPES OF REGI...

JAPAN: ORGANISATION OF PMDA, PHARMACEUTICAL LAWS & REGULATIONS, TYPES OF REGI...
Interactive Powerpoint_How to Master effective communication

Interactive Powerpoint_How to Master effective communication
Ecosystem Interactions Class Discussion Presentation in Blue Green Lined Styl...

Ecosystem Interactions Class Discussion Presentation in Blue Green Lined Styl...
BAG TECHNIQUE Bag technique-a tool making use of public health bag through wh...

BAG TECHNIQUE Bag technique-a tool making use of public health bag through wh...
Periodic table by Cassidy Estabrooke
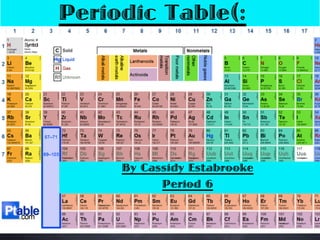
- 1. Periodic Table(: By Cassidy Estabrooke Period 6
- 2. How did the Periodic Table come to be? The discovery of the periodic table goes to Dmitri Mendeleev. Mendeleev developed the periodic table in 1869, where he developed it is unknown.
- 3. How is the table organized? As you can see the Periodic Table is organized like a big grid. The Table is organized into periods going from left to right by increasing atomic number. It is also organized by groups based on what elements share chemical and physical properties.
- 4. How else? There are also two more ways to group the table. Non- Metals Metals
- 5. Relationships? With where the elements lie on the table there is a relationship to the spot they are at. One relationship is that they can be grouped into areas that have similar properties. Another way is that they are grouped in order of increasing atomic mass.
- 6. Calcium! Name: Calcium Atomic number: 20 Element symbol: Ca Atomic Mass: 40.078 How is it used?: Calcium is used in many cellular processes. One main way is that it is essential for living organisms.
- 7. Helium Name: Helium (Helium is named after the greek god of sun Helios.) Atomic number: 2 Element symbol: He Atomic Mass: 4.0026 How is it used?: Helium has many uses but one very interesting one is its use in growing crystals to make silicon wafers.Its a Colorless gas.
- 8. Iron Name: Iron (The element symbol Fe is Latin for ferrum) Atomic number: 26 Element symbol: Fe Atomic Mass: 55.845 How is it used?: Iron forms much of earth's outer and inner core. Iron also comes in many different forms. It is used to make steel.
- 9. The End!(:
Editor's Notes
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
