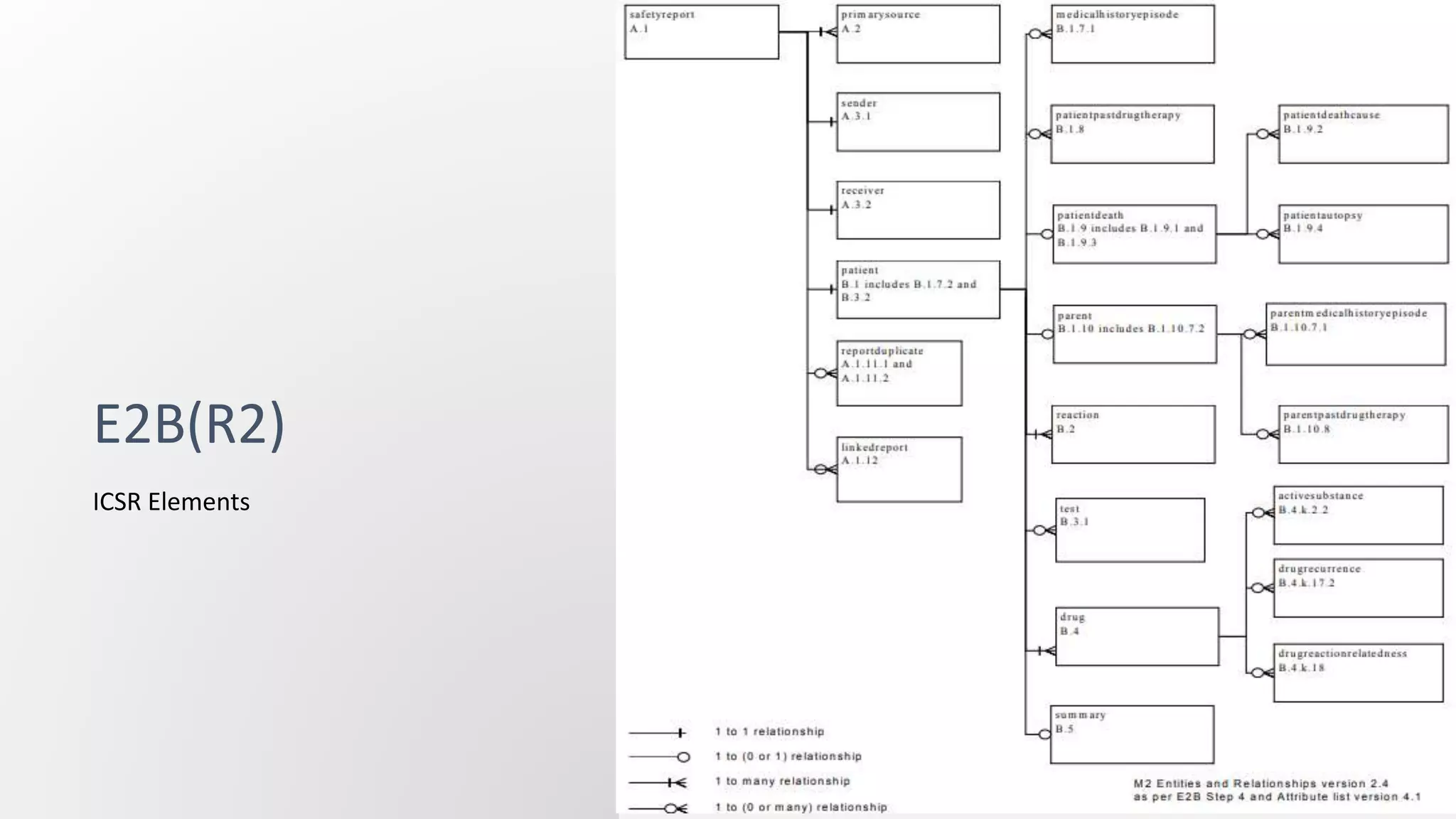
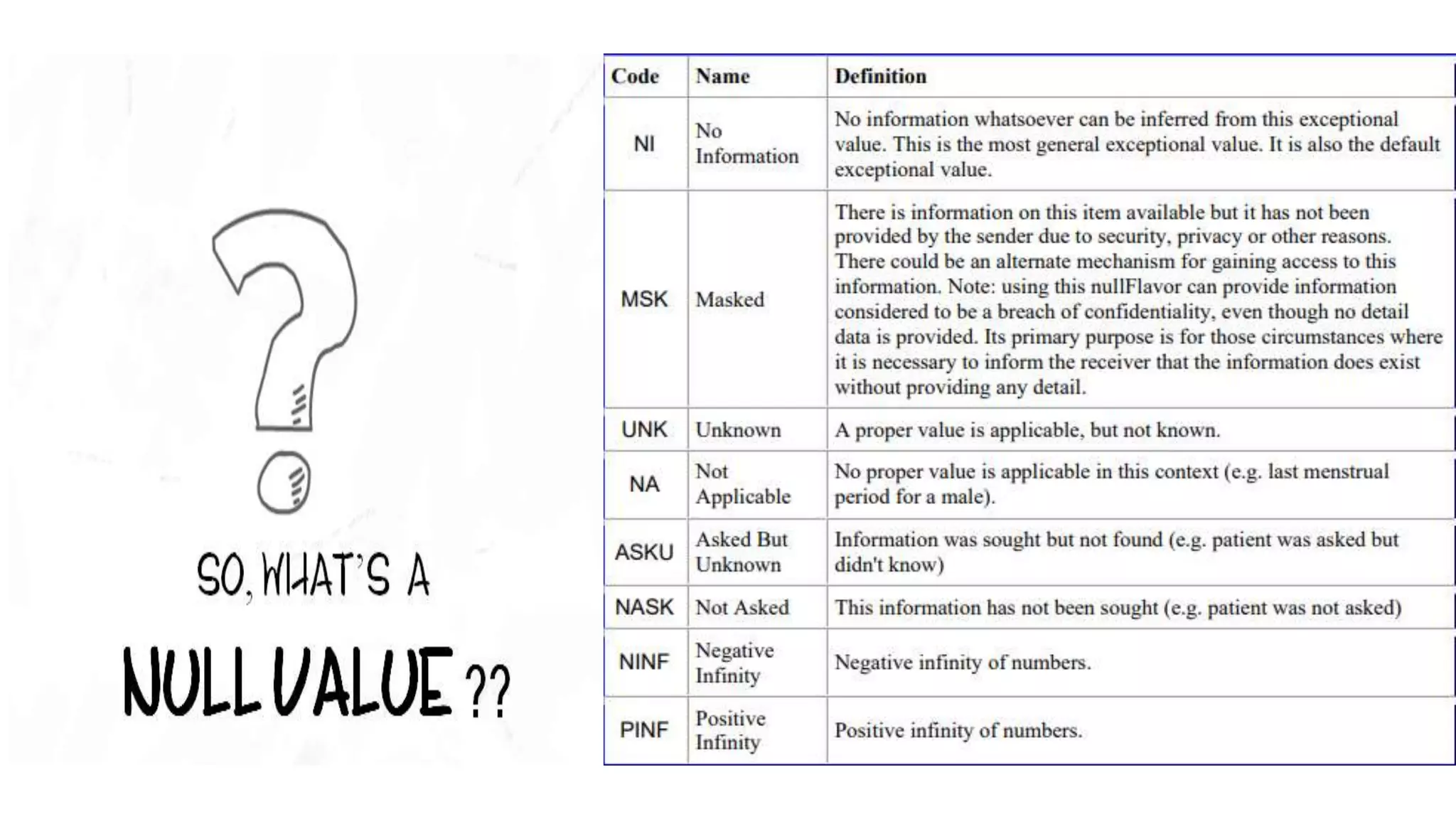
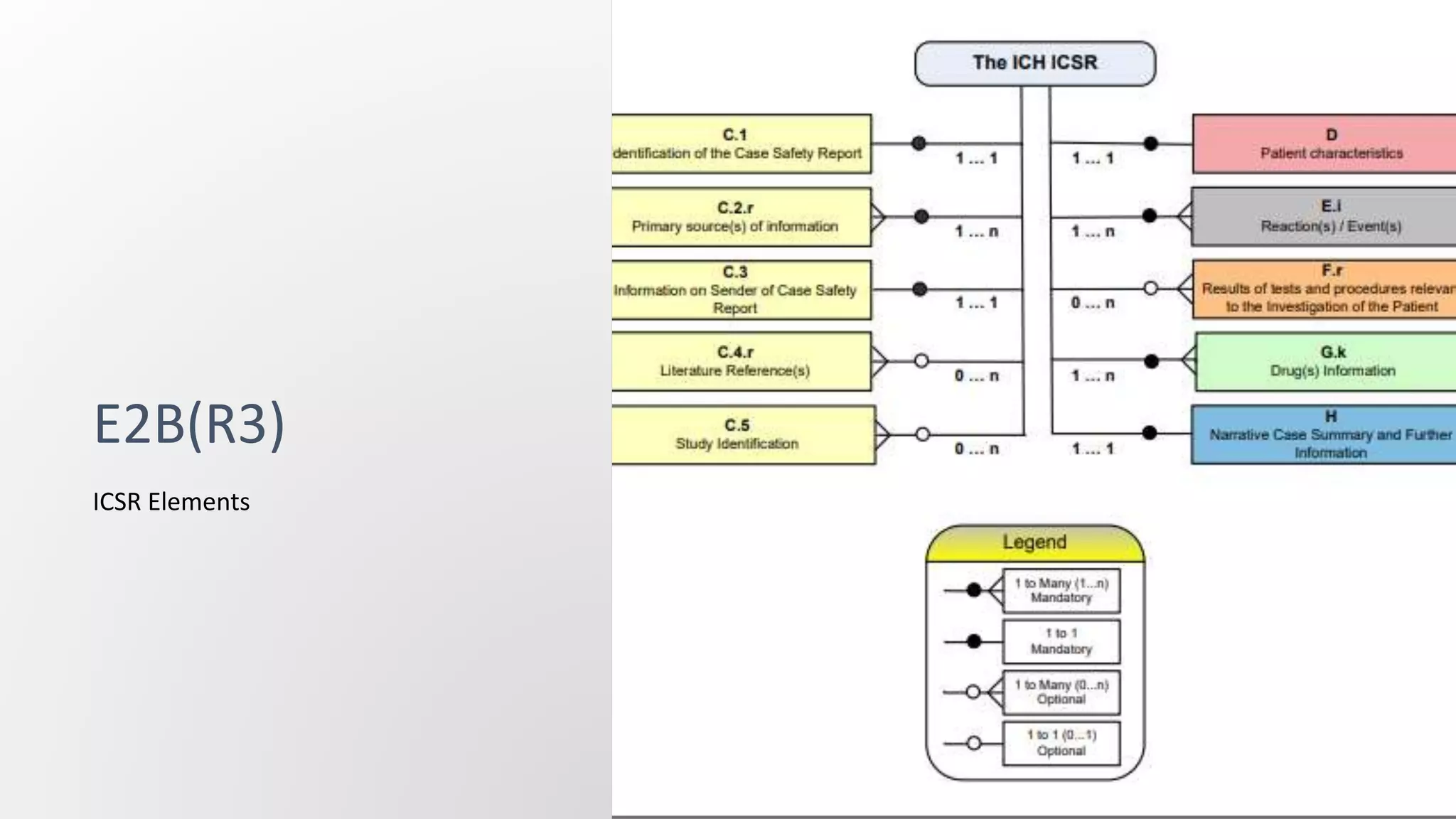
E2B(R2) and E2B(R3) are standards for electronic transmission of individual case safety reports (ICSRs). E2B(R3) was developed through a new collaborative approach between several international organizations, whereas E2B(R2) was developed by ICH alone. E2B(R3) uses a hierarchical data structure and is more standardized and interoperable compared to E2B(R2). The increased standardization of E2B(R3) allows for better exchange of safety information.