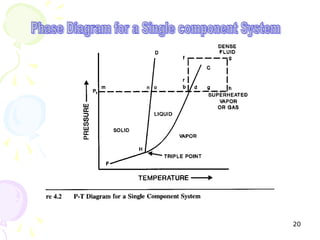
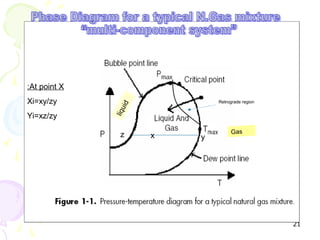
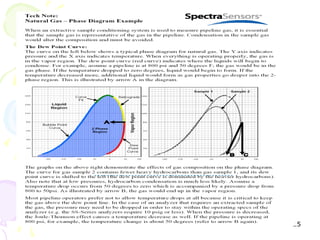
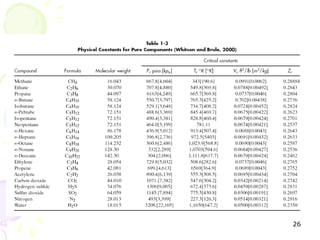
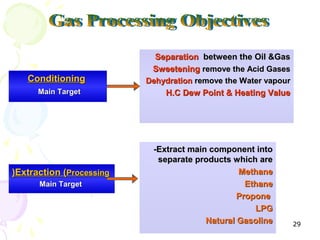
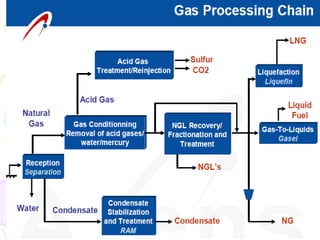
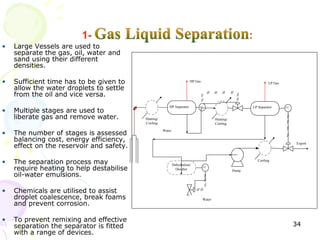
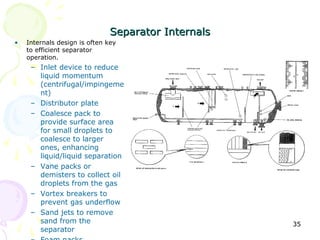
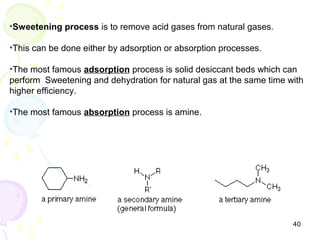
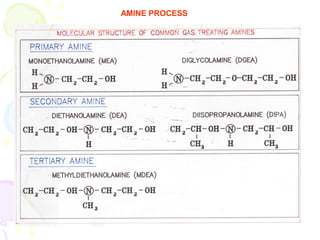
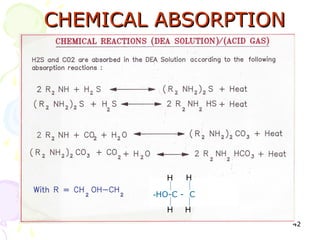
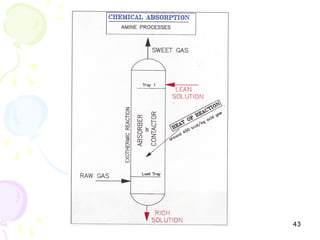
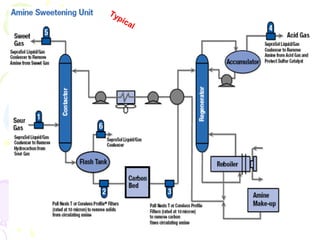
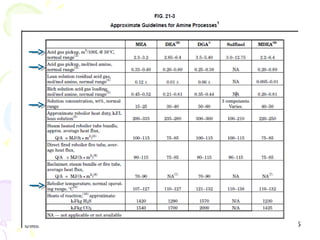
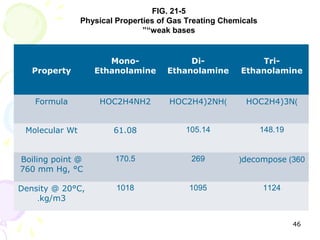
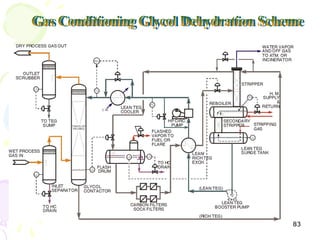
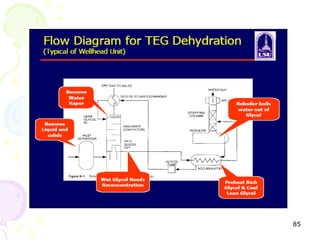
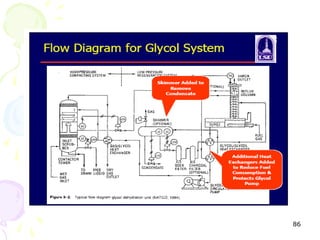
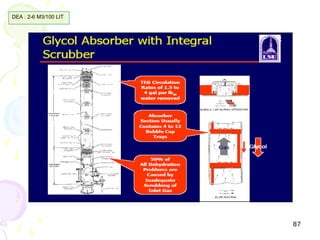
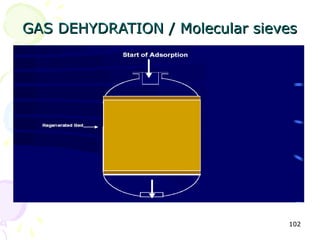
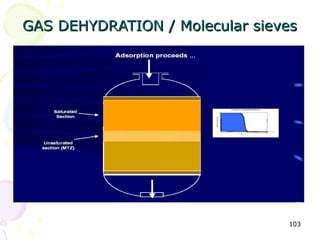
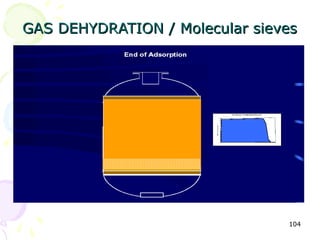
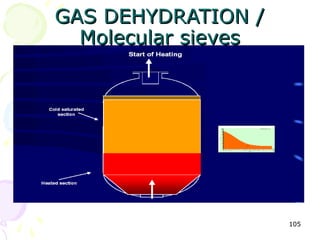
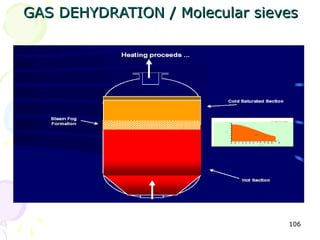
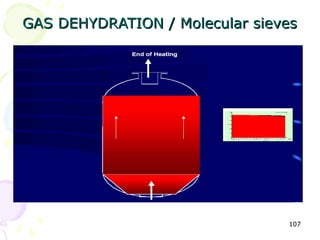
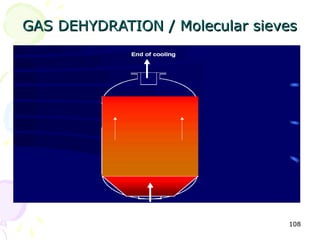
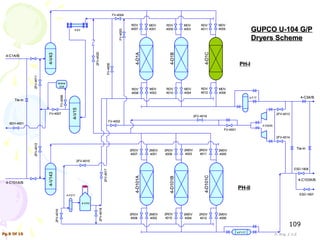
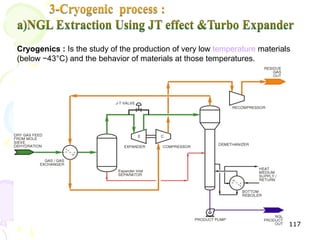
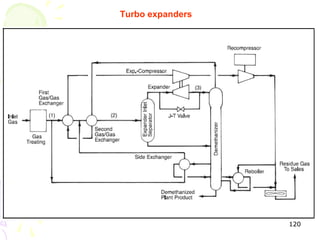
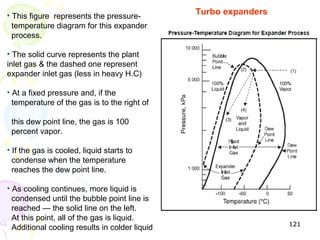
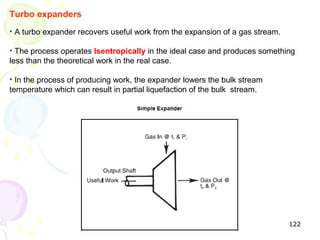
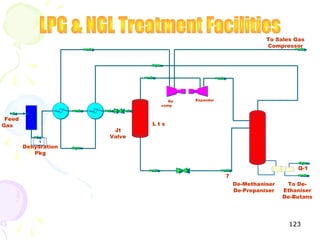
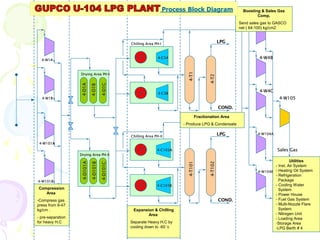
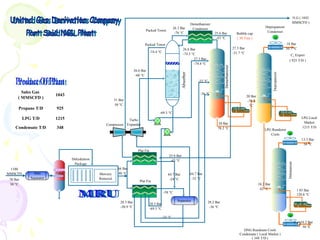
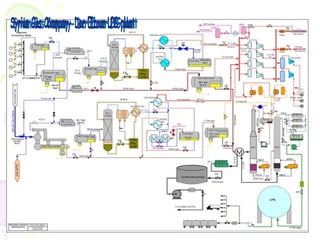
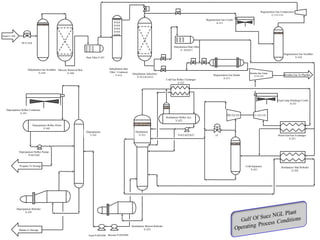
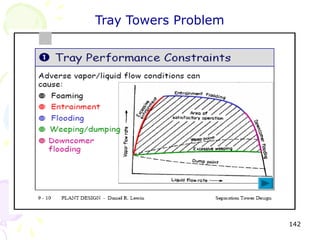
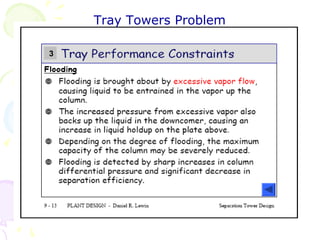
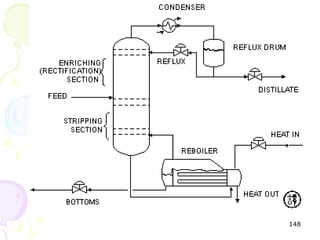
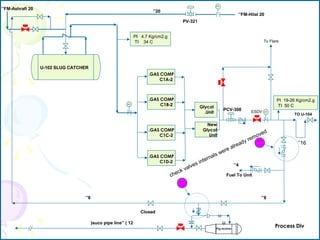
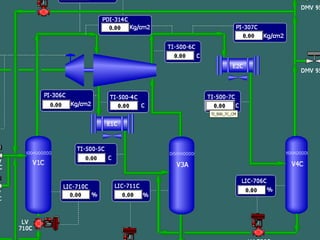
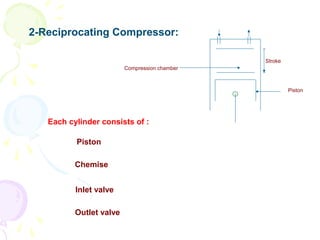
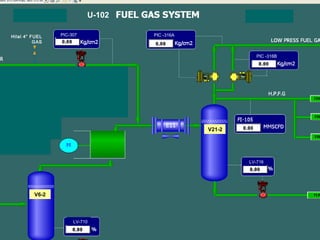
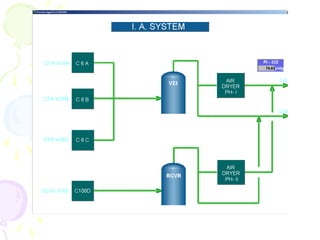
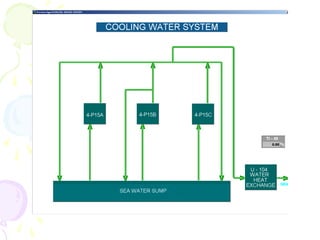
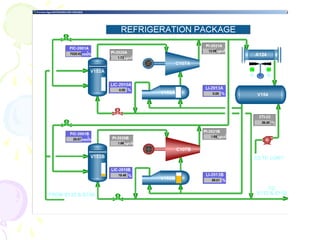
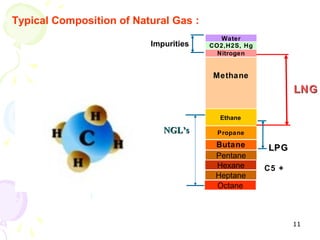
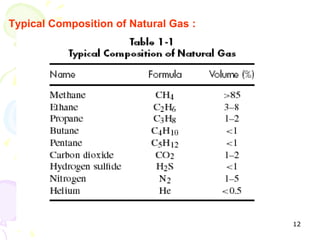
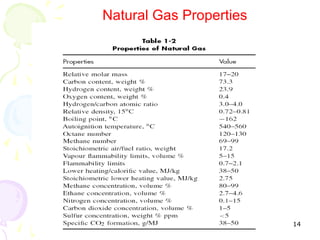
The document provides a comprehensive overview of natural gas, covering its definitions, formation, composition, and properties, as well as various processing methods like gas sweetening and dehydration. It details troubleshooting for amine and glycol units in gas conditioning, along with descriptions of separation techniques and equipment used in gas plants. Additionally, the document discusses the phase behavior of natural gas and definitions of key terms relevant to the industry.












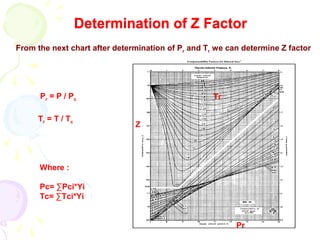
![Behavior of Real Gases
PV = ZnRT
Where :
Z is deviation or compressibility factor and can be expressed as
Z = [ actual volume of n moles of gas / (ideal volume of n moles of gas
at certain P & T) at same P & T ]
where Z is dimensionless.
16](https://image.slidesharecdn.com/n-gasdehydrationboosterstationutilitiesdubai-130315160039-phpapp01/85/NATURAL-GAS-DEHYDRATION-16-320.jpg)