Week 1 a
•Download as PPTX, PDF•
0 likes•184 views
Suspension & Colloids Science 6
Report
Share
Report
Share
Recommended
More Related Content
What's hot
What's hot (20)
Chapter - 2, Is matter around us pure?, Science, Class 9

Chapter - 2, Is matter around us pure?, Science, Class 9
Presentation of mixtures & solutions.pptx by kk shai

Presentation of mixtures & solutions.pptx by kk shai
Similar to Week 1 a
Similar to Week 1 a (20)
kanak garg 9h chem solution,suspension and colloid.pptx

kanak garg 9h chem solution,suspension and colloid.pptx
2ismatterarounduspure-150801135536-lva1-app6891.pdf

2ismatterarounduspure-150801135536-lva1-app6891.pdf
More from YhanzieCapilitan
More from YhanzieCapilitan (20)
Recently uploaded
This presentation was provided by William Mattingly of the Smithsonian Institution, during the fourth segment of the NISO training series "AI & Prompt Design." Session Four: Structured Data and Assistants, was held on April 25, 2024.Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"

Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"National Information Standards Organization (NISO)
APM Welcome
Tuesday 30 April 2024
APM North West Network Conference, Synergies Across Sectors
Presented by:
Professor Adam Boddison OBE, Chief Executive Officer, APM
Conference overview:
https://www.apm.org.uk/community/apm-north-west-branch-conference/
Content description:
APM welcome from CEO
The main conference objective was to promote the Project Management profession with interaction between project practitioners, APM Corporate members, current project management students, academia and all who have an interest in projects.APM Welcome, APM North West Network Conference, Synergies Across Sectors

APM Welcome, APM North West Network Conference, Synergies Across SectorsAssociation for Project Management
God is a creative God Gen 1:1. All that He created was “good”, could also be translated “beautiful”. God created man in His own image Gen 1:27. Maths helps us discover the beauty that God has created in His world and, in turn, create beautiful designs to serve and enrich the lives of others.
Explore beautiful and ugly buildings. Mathematics helps us create beautiful d...

Explore beautiful and ugly buildings. Mathematics helps us create beautiful d...christianmathematics
Recently uploaded (20)
Kisan Call Centre - To harness potential of ICT in Agriculture by answer farm...

Kisan Call Centre - To harness potential of ICT in Agriculture by answer farm...
Presentation by Andreas Schleicher Tackling the School Absenteeism Crisis 30 ...

Presentation by Andreas Schleicher Tackling the School Absenteeism Crisis 30 ...
Interactive Powerpoint_How to Master effective communication

Interactive Powerpoint_How to Master effective communication
Ecosystem Interactions Class Discussion Presentation in Blue Green Lined Styl...

Ecosystem Interactions Class Discussion Presentation in Blue Green Lined Styl...
Russian Escort Service in Delhi 11k Hotel Foreigner Russian Call Girls in Delhi

Russian Escort Service in Delhi 11k Hotel Foreigner Russian Call Girls in Delhi
Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"

Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"
APM Welcome, APM North West Network Conference, Synergies Across Sectors

APM Welcome, APM North West Network Conference, Synergies Across Sectors
Measures of Dispersion and Variability: Range, QD, AD and SD

Measures of Dispersion and Variability: Range, QD, AD and SD
Web & Social Media Analytics Previous Year Question Paper.pdf

Web & Social Media Analytics Previous Year Question Paper.pdf
Explore beautiful and ugly buildings. Mathematics helps us create beautiful d...

Explore beautiful and ugly buildings. Mathematics helps us create beautiful d...
Week 1 a
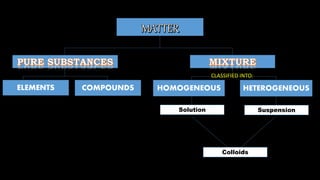
- 1. HOMOGENEOUS HETEROGENEOUSELEMENTS COMPOUNDS Solution Suspension Colloids CLASSIFIED INTO:
- 2. Two-phase mixture, solid is dispersed or spread in solid, liquid and gas. A heterogeneous mixture, components can be distinguished from each other. It is composed of two substances: – the solute in a solution – solvent in a solution Dispersed material (suspension) are larger than the solute in the solution, and they are distributed in dispersion medium.
- 3. Dispersed materials when mixed with dispersion medium makes the suspension turbid. When left alone the dispersed materials becomes visible and settle at the bottom (container). Example: 1. Flour in water 2. Fine pepper in a glass of water 3. Mud in the road
- 4. Example: cake (mixture of flour, sugar and other ingredients), dirt and soil, rice and corn. Example: sand and water, sawdust (lighter particle of sand floating in water), chocolate granules sink at the bottom of the glass of water
- 5. Example: soot and dust suspended in the air Example: water and oil SOOT POLLUTION WATER AND OIL
- 6. like baking. such as minute maid (with pulp and bits). (such as water and sand) in the process of purifying water. – liquid suspension for the kids.
- 7. Homogeneous mixture that sometimes appears as heterogeneous mixture. A mixture of: 1. Dispersed phase – solute in the solution. It can be solid, liquid, or gas. The size of the substance is in between those in solution and a suspension. 2. Dispersion medium – solvent in the solution. It can also be solid, liquid, or gas.
- 8. – dispersed phase and dispersed medium are both solid. Example: gemstones, pearls, and colored glasses – result from dispersing liquids in a solid medium. Example: jelly, jam, cheese, butter, and gelatin – dispersed phase is a gas and the dispersion medium is solid. Example: rubber bands, pumice, Styrofoam GELATIN PUMICE ROCK
- 9. – solids are dispersed in liquid medium. Example: paint, milk of magnesia, and muddy water – formed when both the dispersed phase and dispersion medium are liquids. Example: milk, hair cream, mayonnaise, – when gas dispersed in a liquid medium Example: froth, soap lather, whipped cream Milk of magnesia Froth Soap lather
- 10. – colloids of gas. There are solid and liquid aerosols. Example: 1. smoke (fine solid particles dispersed in gas); 2. fog (liquid substances dispersed in gas ); 3. soot and dispersed dust (solid aerosols); 4. insecticides and hair spray mists ( liquid aerosols); and 5. clouds (tiny water droplets dispersed in air)
- 11. – making of dough into bread. – gelatin, jelly and whipped cream. – hair spray – pen ink – paints, dyestuff, paper and lubricant