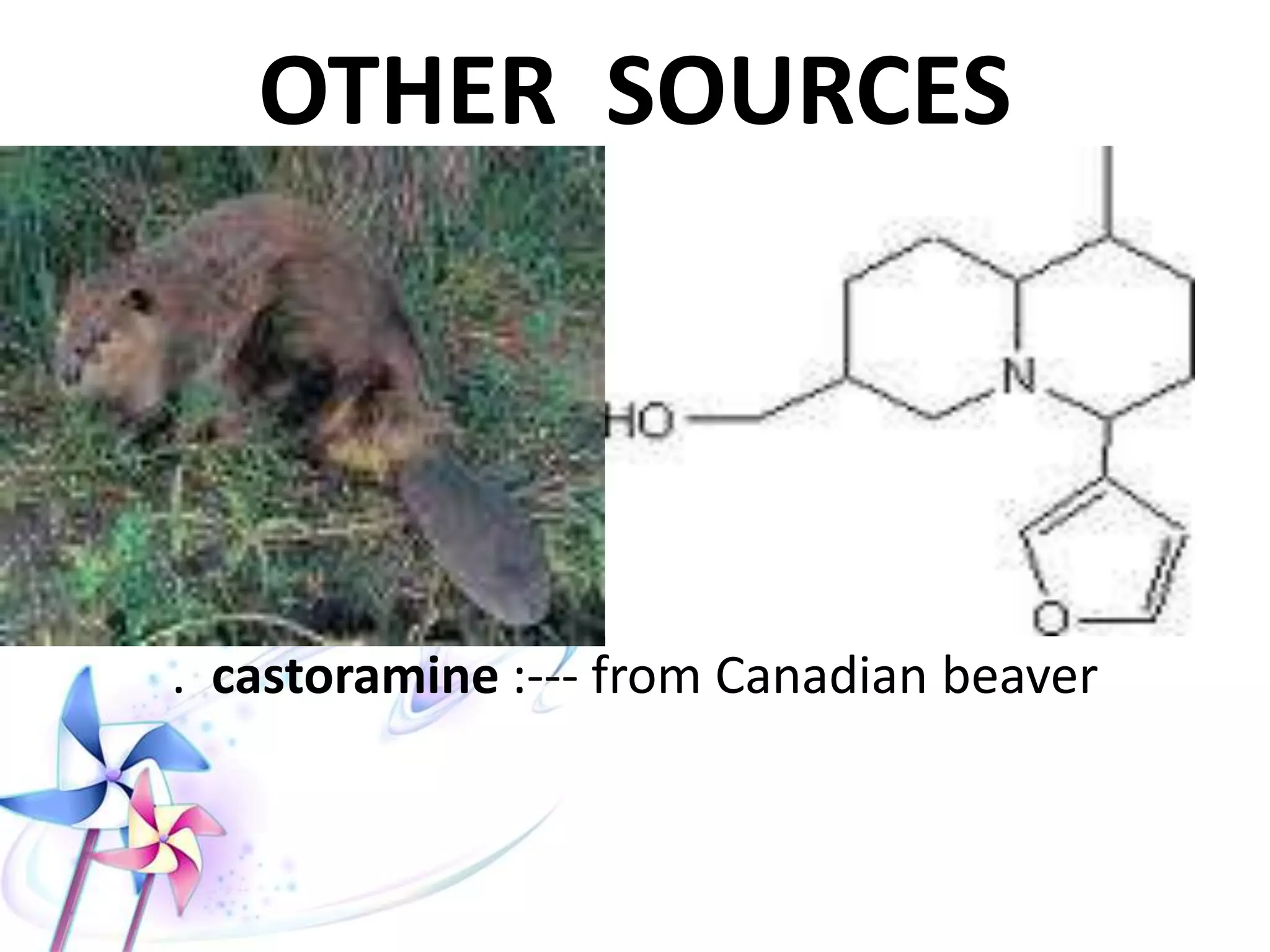
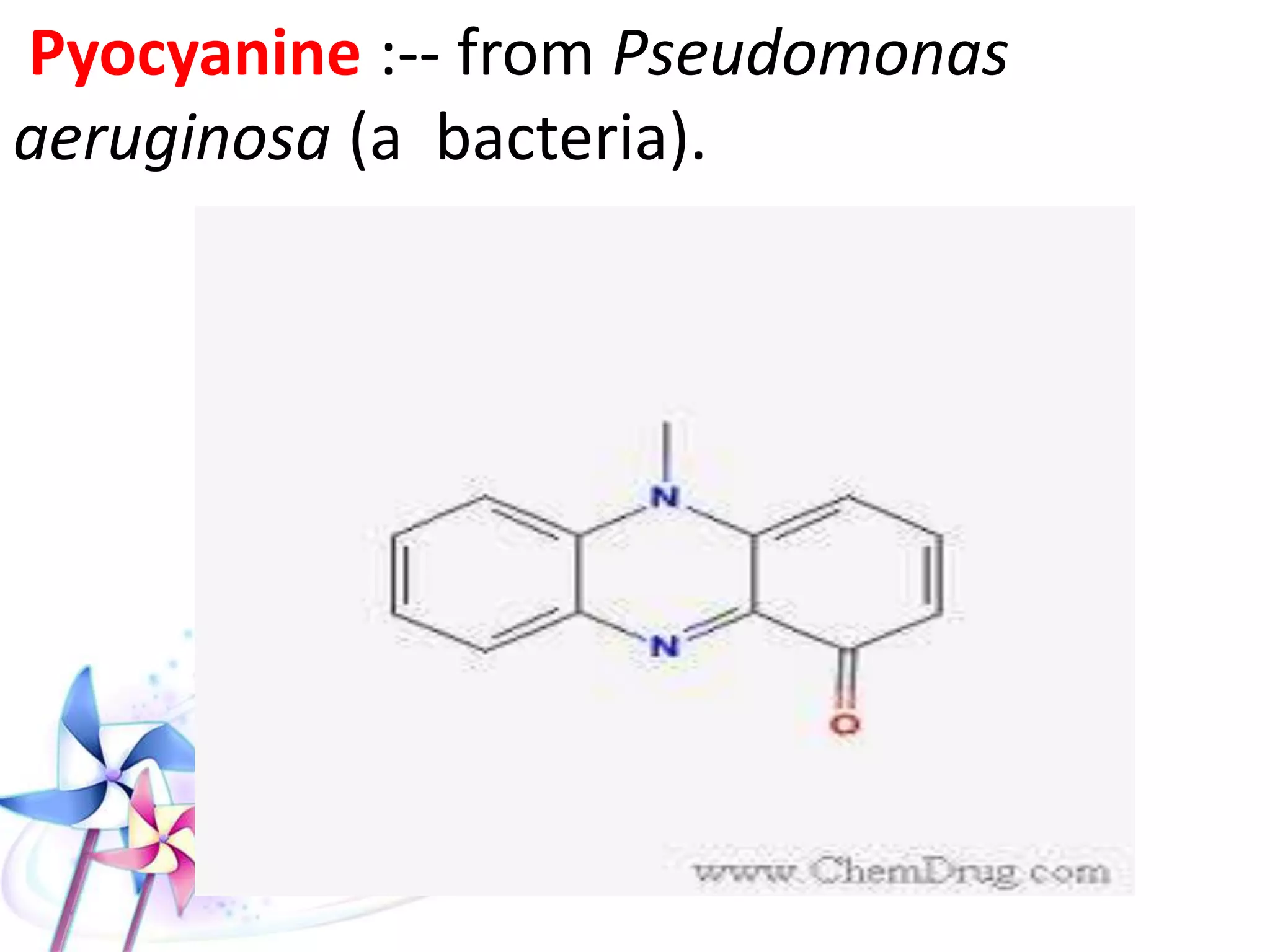
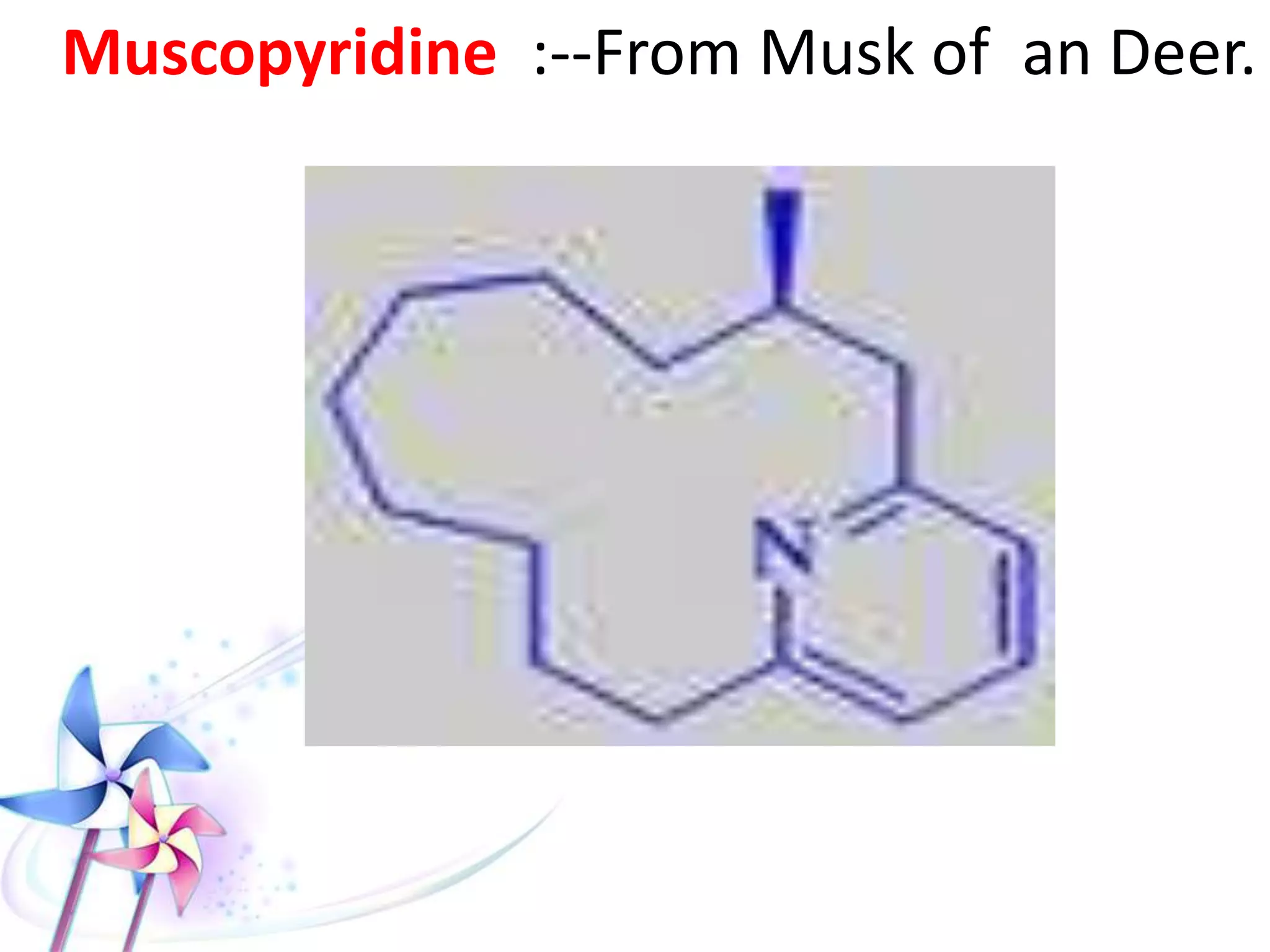
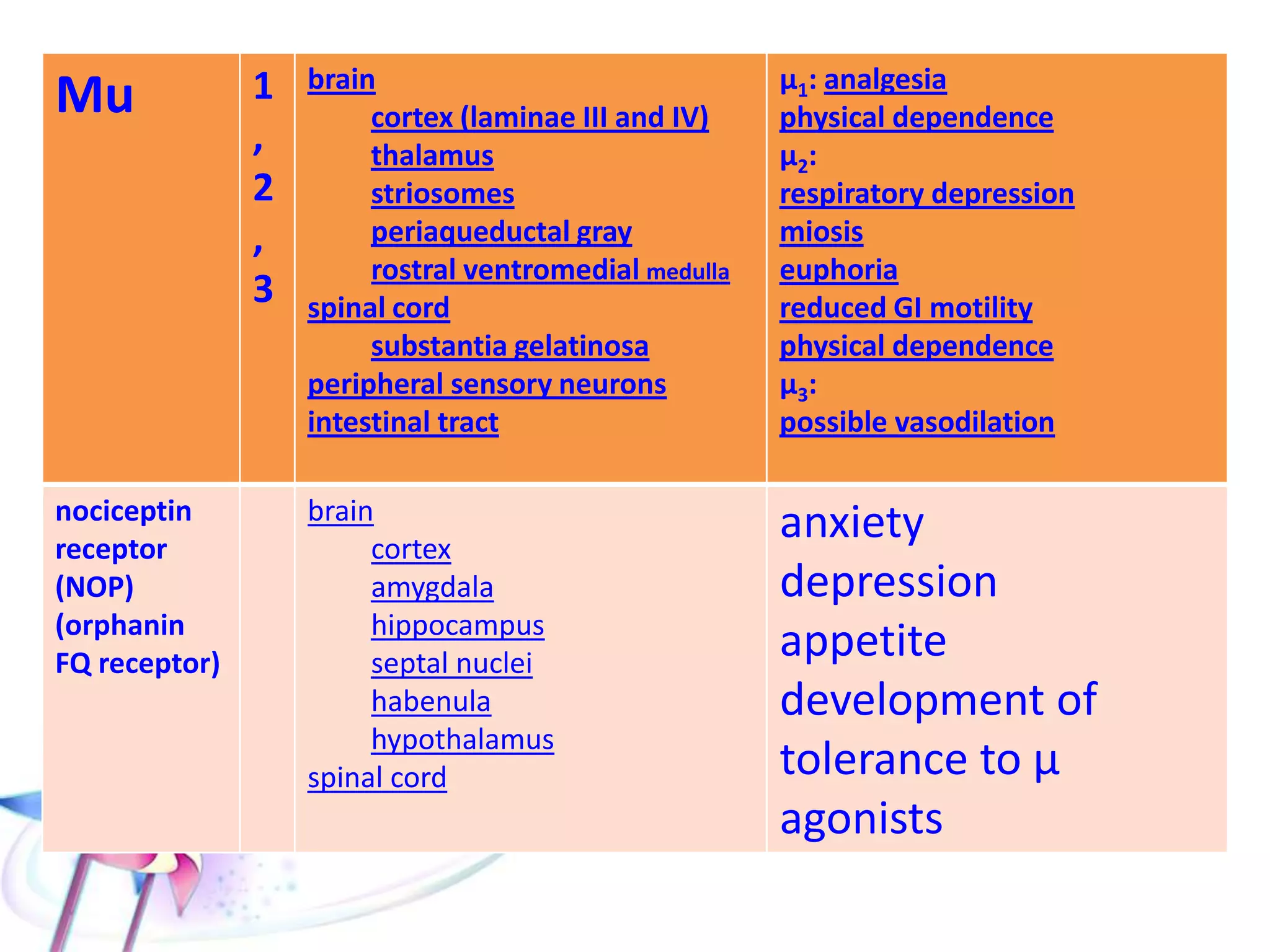
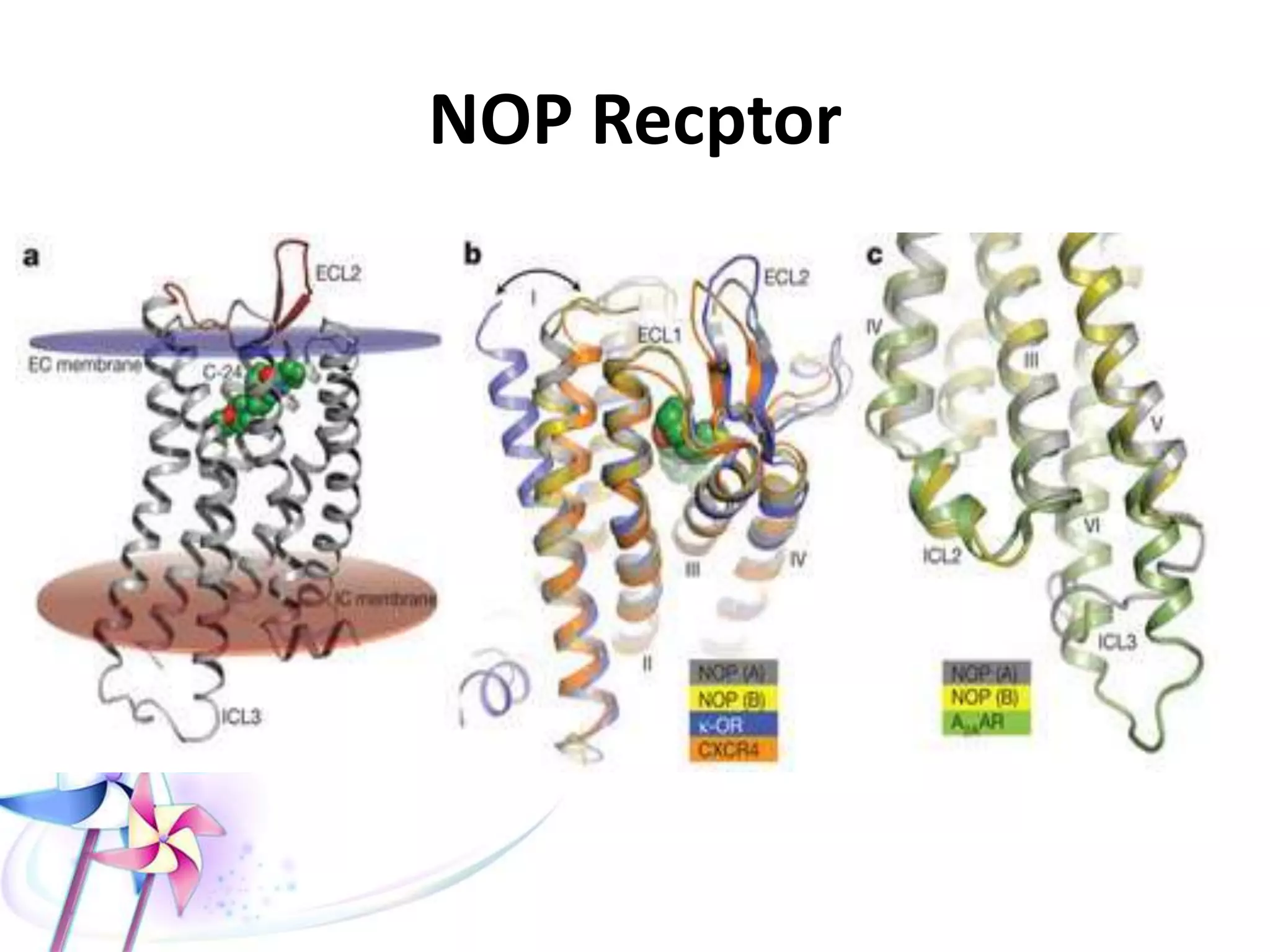
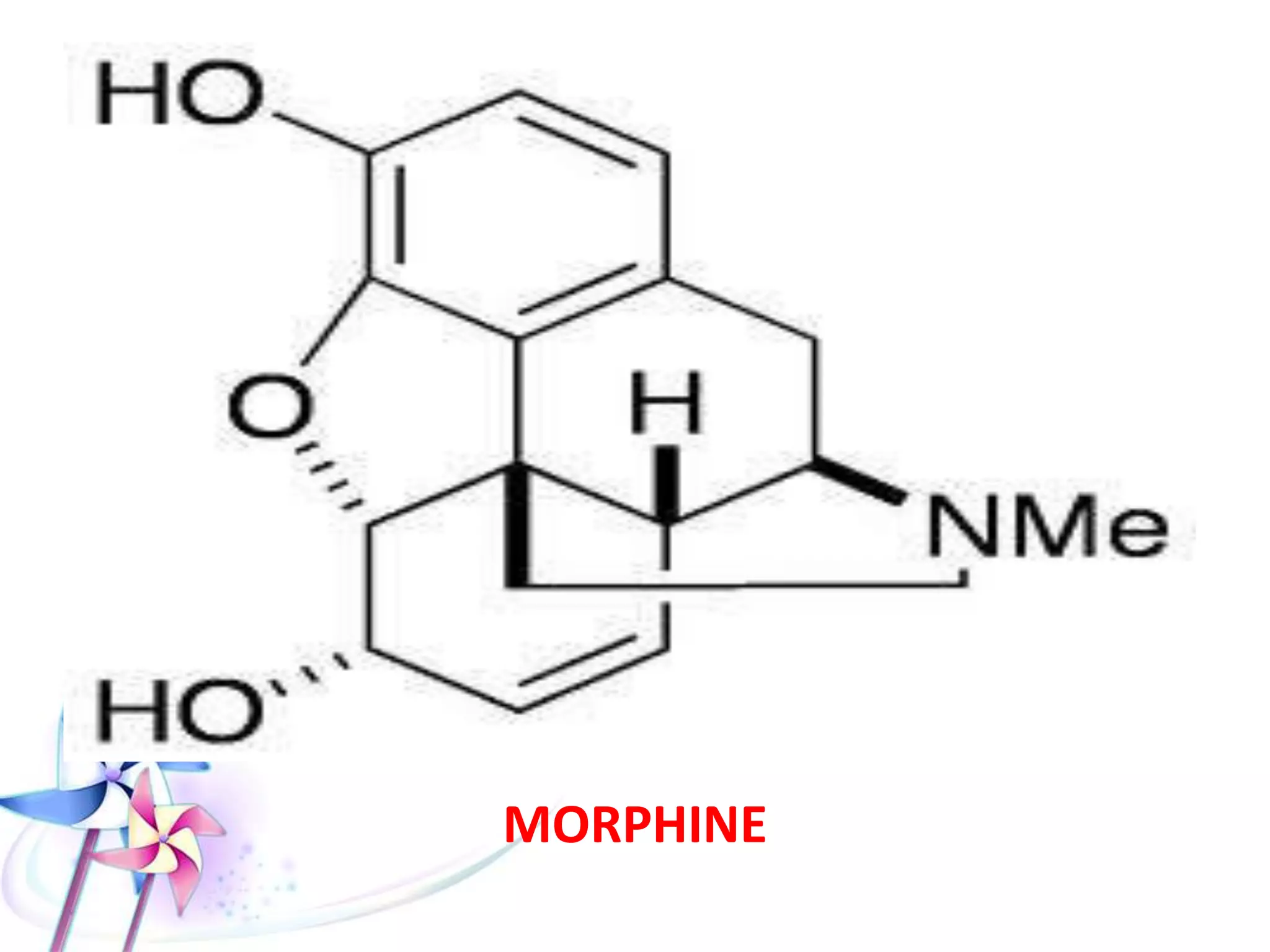
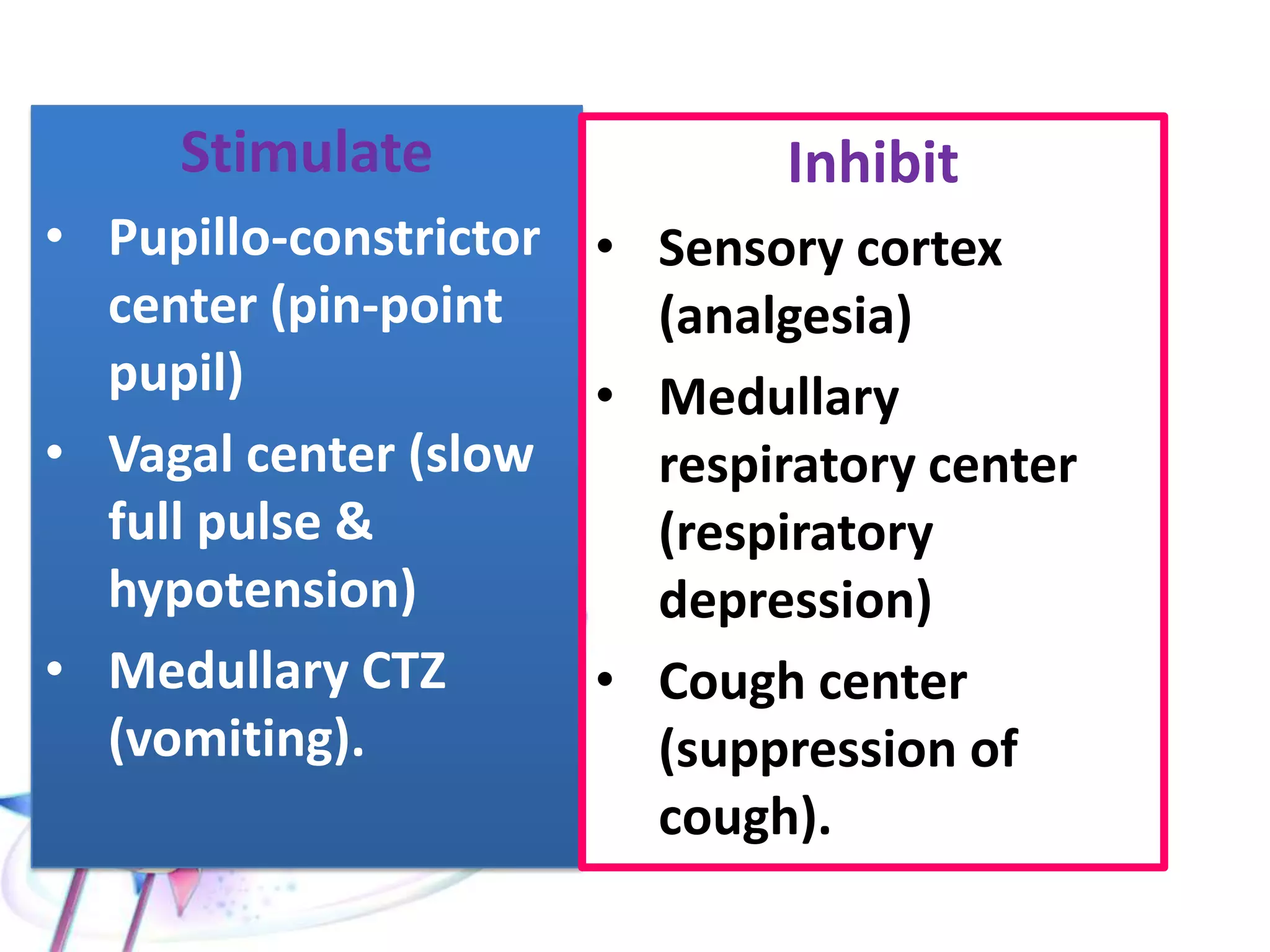
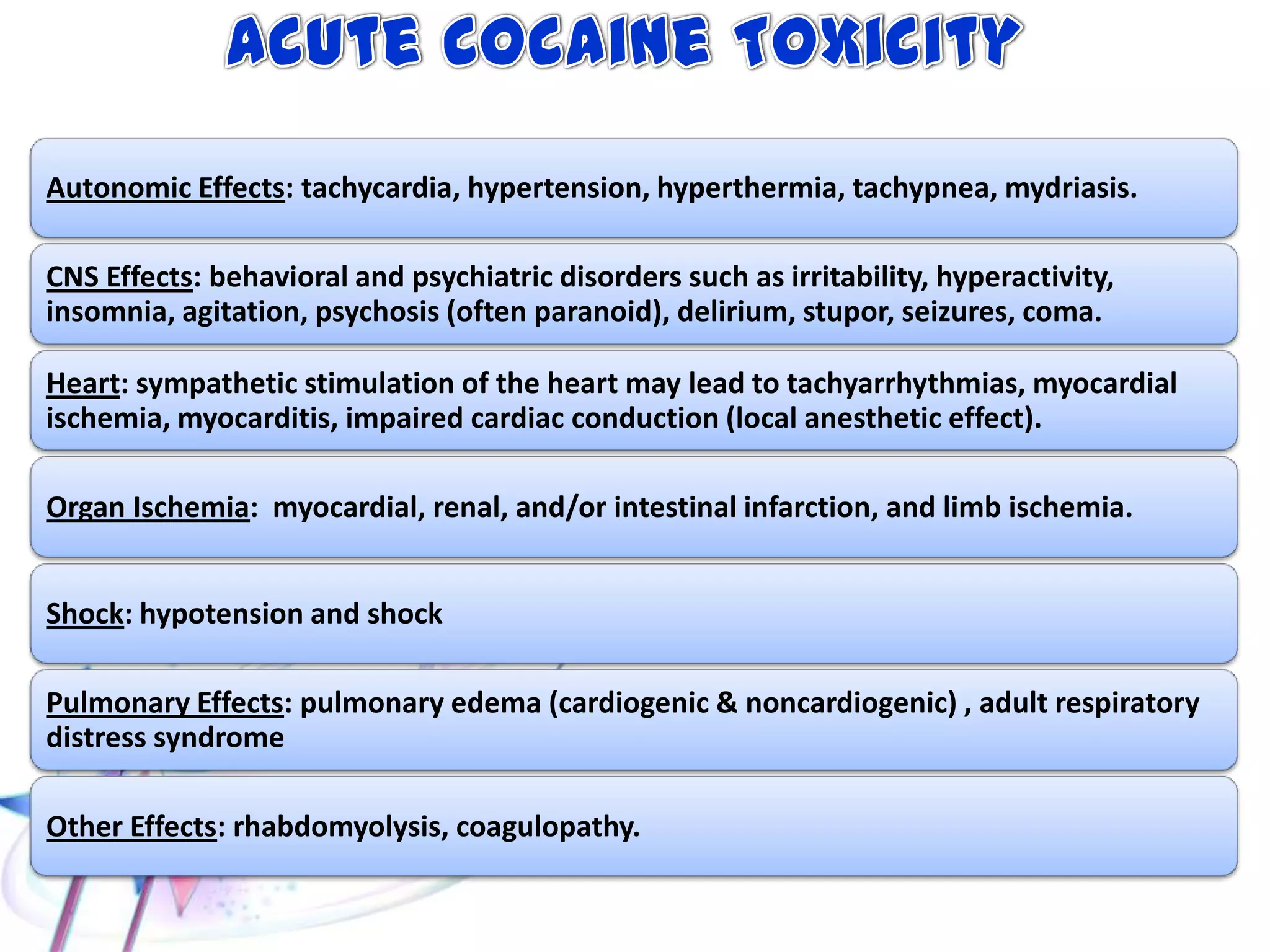
This document discusses the toxicity of plant alkaloids. It begins by defining plant alkaloids as basic heterocyclic nitrogenous compounds of plant origin that are physiologically active. It notes some deviations from this definition, and discusses other non-plant sources of alkaloids. The document then classifies alkaloids and discusses some of the major types including opioids, opiates, and their receptors. Specific alkaloids like morphine, heroin, cocaine, and cannabis are discussed in more detail regarding their sources, effects, toxicity, and clinical manifestations of overdose.