Some Facts about Urea Stripper By Prem Baboo.pdf
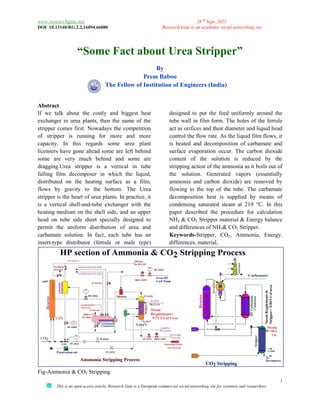
- 1. www.researchgate.net DOI: 10.13140/RG.2.2.16894.66880 This is an open access article, Research Gate is a European commercial social networking site for scientists and researchers “Some Fact about T The Fellow of Institution of Engineers (India) Abstract If we talk about the costly and exchanger in urea plants, then the name of the stripper comes first. Nowadays the competition of stripper is running for more and more capacity. In this regards some urea plant licensers have gone ahead some are left behind some are very much behind and some are dragging.Urea stripper is a vertical in tube falling film decomposer in which the liquid, distributed on the heating surface as a film, flows by gravity to the bottom. stripper is the heart of urea plants. is a vertical shell-and-tube exchanger with the heating medium on the shell side, and an upper head on tube side sheet specially designed to permit the uniform distribution of urea and carbamate solution. In fact, each tube has an insert-type distributor (ferrule or male type Fig-Ammonia & CO2 Stripping 28 th Sept, 2022 Research Gate is an academic social networking This is an open access article, Research Gate is a European commercial social networking site for scientists and researchers Some Fact about Urea Stripper” By Prem Baboo Fellow of Institution of Engineers (India) costly and biggest heat , then the name of the Nowadays the competition of stripper is running for more and more capacity. In this regards some urea plant licensers have gone ahead some are left behind behind and some are is a vertical in tube falling film decomposer in which the liquid, distributed on the heating surface as a film, flows by gravity to the bottom. The Urea In practice, it e exchanger with the heating medium on the shell side, and an upper head on tube side sheet specially designed to permit the uniform distribution of urea and carbamate solution. In fact, each tube has an or male type) designed to put the feed uniformly around the tube wall in film form. The holes of the ferrule act as orifices and their diameter and liquid head control the flow rate. As the liquid film flows, it is heated and decomposition of carbamate and surface evaporation occur. The carbon dioxide content of the solution is reduced by the stripping action of the ammonia as it boils out of the solution. Generated vapors (essentially ammonia and carbon dioxide) are removed by flowing to the top of the tube. The carbamate decomposition heat is supplied by means of condensing saturated steam at 219 °C. paper described the procedure for calculation NH3 & CO2 Stripper material & Energy balance and differences of NH3& CO2 Keywords-Stripper, CO2, Ammonia, differences, material, Research Gate is an academic social networking site 1 This is an open access article, Research Gate is a European commercial social networking site for scientists and researchers ” ned to put the feed uniformly around the tube wall in film form. The holes of the ferrule act as orifices and their diameter and liquid head control the flow rate. As the liquid film flows, it is heated and decomposition of carbamate and n occur. The carbon dioxide content of the solution is reduced by the stripping action of the ammonia as it boils out of the solution. Generated vapors (essentially ammonia and carbon dioxide) are removed by flowing to the top of the tube. The carbamate composition heat is supplied by means of condensing saturated steam at 219 °C. In this procedure for calculation Stripper material & Energy balance Stripper. , Ammonia, Energy.
- 2. www.researchgate.net 28 th Sept, 2022 DOI: 10.13140/RG.2.2.16894.66880 Research Gate is an academic social networking site 2 This is an open access article, Research Gate is a European commercial social networking site for scientists and researchers Fig- Ammonia stripping process Description Efforts to find some additional driving force beyond the usual addition of heat and reduction in pressure to decompose carbamate have met with a great success of totally new route. In this process, NH3 or CO2 is used to strip urea reactor effluent for decomposition of carbamate. The stripper and the reactor are operating essentially at the same pressure of about 140 to 158 kg/cm2 g. The synthesis mixture from the reactor, consisting of urea, unconverted ammonium carbamate, excess ammonia and water is fed to the top of the stripper. The stripper has two functions i.e. its upper part is equipped with trays where excess ammonia is partly separated from the stripper feed by direct counter current contact of the feed solution with the gas coming from the lower part of the stripper. This pre stripping in the top is said to be required to achieve effective CO2 stripping in the lower part. In the lower part of the stripper (a falling film heater), ammonium carbamate is decomposed and the resulting CO2 and NH3 as well as the excess ammonia are evaporated by CO2 stripping and steam heating. The overhead gaseous mixture from the top of the stripper is introduced into the carbamate condensers. Here, two units in parallel are installed, where the gaseous mixture is condensed and absorbed by the carbamate solution coming from the medium pressure recovery stage. Heat liberated in the high pressure carbamate condensers is used to generate low pressure steam in one of the condensers and to heat the urea solution from the stripper after the pressure is reduced to about 18- 24 kg/cm2 g in the shell side of the second carbamate condenser. The gas and liquid from the carbamate condensers are recycled to the reactor by gravity flow. Theory of stripping The theory of stripping is based on Henry’s law which states that the concentration of components in a solution while in equilibrium with vapour phase is directly proportional to the partial pressure of the components in vapour phase. If the partial pressure of the one component is altered, while keeping the total pressure constant, the concentration of that component in solution will vary accordingly. This is illustrated as below . Let us assume that A and B are two components.
- 3. www.researchgate.net 28 th Sept, 2022 DOI: 10.13140/RG.2.2.16894.66880 Research Gate is an academic social networking site 3 This is an open access article, Research Gate is a European commercial social networking site for scientists and researchers CA = Concentration of A in solution. CB = Concentration of B in solution. PA = Partial pressure of A in vapour phase PB = Partial pressure of B in vapour phase. CA = k PA, CB =k PB In the stripper which operates at around 140 kg/cm2 a pressure, the carbamate solution is split into it’s components ammonia and CO2 by adopting the above principle. The reactor effluent is counter currently contacted with fresh CO2 which increases the partial pressure of CO2, thereby upsetting the solution concentration and splitting the carbamate into ammonia and CO2. The above is achieved by providing external heat and without change in operating pressure. Technical realization of the stripping process In this process, the formation of urea, the stripping and the condensation of the carbamate are accomplished at the same pressure level. This gives rise to several advantages such as :- Elimination of pumping concentrated carbamate solution from various pressure levels. Minimizing the quantity of water introduced to the reaction system by means of recycle streams. Optimum heat balance with the export of LP steam. Reduced power consumption as reaction pressure is comparatively low. Corrosion is the function of Temperature as well as Corrosiveness of process fluid is not constant owing to effect of start-up/shut-down, fluctuation of temperature and dissolved oxygen content and geometrical flow distribution. For stripper. Passivation air role is principal in this issue The Passivation given in CO2 depends upon the process to process in M/Saipem it is 0.25% and in M/S Stamicarbon it is 0.6%.This passivation depends upon N/C ratio in Reactors. Stripper top and bottom temperature, Design for Saipem 205/190 but after losing of ferrules- 205/192-196.Mega bond stripper is the latest stripper. Average corrosion rate of tubes. The corrosion rate is depends upon Temperature and passivation oxygen, No of shut down etc. Generally Corrosion rate as follows as const. Temperature Conditions. In Zr-0.005 mm/year, Ti-0.05 mm/Year,2 RE-69-0.25 mm/year, corrosion rate in duplex material is about 0.05 mm to 0.1 mm per year.* Stripper Tube diameter-2.7 mm t0 3.5 mm. Difference Between Stripper and Decomposer Sr. No. Parameters Decomposer Stripper 1 Definition “The process based on the 1st principal of decrease in pressure and increase in temperature” and then have a series of decomposition stage where the Reactor discharge is treated in successively at lower pressure. “To reduce the partial pressure of product by swamping the system by one of the reactant which reduces the partial pressure of other reactant considerable without changing the total pressure either CO2 or NH3 or both can be used as a stripping agent.” 2 Delta P between Reactor and stripper Delta P is more than Stripper No Delta between Reactor and Stripper, however in Saipem process there is small delta P. 3 Base of Decomposition Differential pressure solution flashed due to high difference of pressure. Stripping base hennery law of partial pressure, Mass transfer also involve. Base on partially pressure, In Stamicarbon CO2
- 4. www.researchgate.net 28 th Sept, 2022 DOI: 10.13140/RG.2.2.16894.66880 Research Gate is an academic social networking site 4 This is an open access article, Research Gate is a European commercial social networking site for scientists and researchers introduced in stripper, is advantageous because CO2 increase P1 CO2 to P2 CO2, so P1 NH3 will reduced to P2 NH3 to maintain total pressure constant as PCO2+PNH3=Total Pressure. Now K-1 = (X2NH3X XCO2)/X Carb. at particular temp K1 is constant so when XNH3 is reduced to keep K1 constant X Carbamate will be reduced much faster by decomposition as XNH3 appears in the equation with power of 2 4 Theory Decomposition base only high temp & low pressure, decomposition due to let down water recycle is more. Decomposition is favoured by low pressure but as decomposition products are to be recycled back to reactor it will require energy for this step. Also at low pressure more water is evaporate during decomposition and this water would enter the urea reactor along with recycle stream and will adversely affect the conversion. Thus if the decomposition is carried out in single stage near atmospheric pressure the Carbamate formed during recovery at the same pressure carry a lot of water. considering these factors, decomposition is carried out number of stages. In stripping process the 1st stage decomposition and recovery is done at the reactor pressure which permit heat to be recovered at high level and also results in saving the power for returning the recycle streams to the reactor .however in Saipem process there is difference in pressure in Reactor and Stripper, so additional advantages of deferential decomposition.
- 5. www.researchgate.net DOI: 10.13140/RG.2.2.16894.66880 This is an open access article, Research Gate is a European commercial social networking site for scientists and researchers Fig-Stripper Vs decomposer water carryover Comparison of Ammonia & CO2 Stripper Sr. No. Parameters Ammonia stripping Process 1 Surface Area Less 2 Nos of Tubes Less 3 Cost Less(Except Mega bond) 4 In Case of Tube or tube sheet Leakage In case of Leakage the pH of Steam condensate is more than 9.0 ammonia content is higher Hence no problem of corrosion, you can run the plant at minor leakage by separate draining of condensate, 5 Steam Requirement 0.72 T/ton of Urea 28 th Sept, 2022 Research Gate is an academic social networking This is an open access article, Research Gate is a European commercial social networking site for scientists and researchers Stripper Vs decomposer water carryover Stripper Ammonia stripping Process CO2 Stripping Process More in same plant Load because the double load of heating(CO2 as well as Reactor effluents) About 150 % tubes more (Except Mega bond) Very Costly at same plant load In case of Leakage the pH of Steam condensate is more than 9.0-10.0 because the ammonia content is higher, Hence no problem of corrosion, you can run the plant at minor leakage by separate draining of condensate, In case of minor leakage , take shut down immediately because the pH of steam condensate come down below 7.0 due to CO2 contents is higher and hence corrosion start in CS shell, otherwise equipment will damage and cause major incident. T/ton of Urea 0.84 T / ton of urea Research Gate is an academic social networking site 5 This is an open access article, Research Gate is a European commercial social networking site for scientists and researchers because the double as well as Reactor Very Costly at same plant load In case of minor leakage , take shut down mmediately because the pH of steam condensate come down below 7.0 due to and hence corrosion start in CS shell, otherwise equipment will damage and cause major incident.
- 6. www.researchgate.net DOI: 10.13140/RG.2.2.16894.66880 This is an open access article, Research Gate is a European commercial social networking site for scientists and researchers 6 Stripper inlet Temp, 0 C 7 Stripper outlet Temp, 0 C 204(211 Mega 8 Stripper vapour Temp, 0 C 9 CO2 to stripper Temp 0 C 10 O2 in CO2 % Fig-mega bond tube 28 th Sept, 2022 Research Gate is an academic social networking This is an open access article, Research Gate is a European commercial social networking site for scientists and researchers 188 185 (211 Mega bond) 176 190 189 NA 120 0.25 0.6 The Omega bond stripper is so costly, everyone can dare. Replacing the old stripper in old unit is one choice. In the Titanium stripper bottom temperature easily maintain 210 without any problem ig ignore some problem like tube end corrosion. This 210 temperature at which challenging all the process we can run the plant at any load. The ideal temperature for urea stripping is between 210 – 212˚C However, due to the upper temperature limits of stainless steel, plants cannot operate at their optimum capabilities, sacrificing significant plant capacity. Passivation air is the main factor in the urea process on which the plant safety explosion etc and losses also depends. Passivation air must also be removed after stripping. This adds process costs and hazards resulting from the co oxygen and hydrogen—an explosive gas mixture the mega bond is the solution of all problems this is the mother/father of urea plants. Passivation air is not necessary with titanium and zirconium. Eliminating the need to remove passivation air downstream improves yield, Research Gate is an academic social networking site 6 This is an open access article, Research Gate is a European commercial social networking site for scientists and researchers The Omega bond stripper is so costly, not everyone can dare. Replacing the old stripper in old unit is one choice. In the Titanium stripper bottom temperature easily maintain 2100 C without any problem ig ignore some problem like tube end corrosion. This 2100 C is the enging all the process we can run the plant at any load. The ideal temperature for urea stripping is between 210˚C ˚C However, due to the upper temperature limits of stainless steel, plants cannot operate at their optimum capabilities, sacrificing nificant plant capacity. Passivation air is the main factor in the urea process on which the plant safety explosion etc and losses also depends. Passivation air must also be removed after stripping. This adds process costs and hazards resulting from the combination of an explosive gas mixture the mega bond is the solution of all problems this is the mother/father of urea plants. Passivation air is not necessary with titanium and zirconium. Eliminating the need to remove ownstream improves yield,
- 7. www.researchgate.net 28 th Sept, 2022 DOI: 10.13140/RG.2.2.16894.66880 Research Gate is an academic social networking site 7 This is an open access article, Research Gate is a European commercial social networking site for scientists and researchers reduces environmental concerns and improves plant safety. In addition, next-generation urea tubing eliminates temperature limitations inherent with steel alternatives, allowing the strippers to run at 212°C, thus improving capacity by up to 15 percent. In addition, corrosion is eliminated in the stripper, ensuring impurities such as iron, nickel and chrome—the core components of stainless steel—do not exist in the final product. Erosion inside the tubes is also eliminated, ensuring an extensive stripper life without the need to flip the stripper. Next- generation urea tubing works with standard titanium stripper designs, replacing pure titanium tubes. Omega Bond tubing is the next generation solution; it is the only technology that combines the benefits of Zirconium and Titanium, ideally optimizing urea processing. ATI is a world leader in specialty metal manufacturing, with over 50 years of experience from raw material to final product ensuring a reliable and high-quality solution. Omega Bond Tubing combines titanium grade 3 and Zirconium 702 using a unique metallurgical process that creates high-quality corrosion- and erosion-resistant tubing designed to leverage the strengths of each metal. The result: maintenance-free operations, less downtime and higher quality output.
- 8. www.researchgate.net DOI: 10.13140/RG.2.2.16894.66880 This is an open access article, Research Gate is a European commercial social networking site for scientists and researchers Fig- Comparison of stripping process Compression of NH Operating Feutures Particular Fluid circulated Total fluid entering Fluid vaporised or condensed Kg/Hr Temperature in Temperature out Operating pressure kg/cm Design pressure kg/cm Design temperature Table –Comparison of stripper 28 th Sept, 2022 Research Gate is an academic social networking This is an open access article, Research Gate is a European commercial social networking site for scientists and researchers Comparison of stripping process Compression of NH3 & CO2 Stripper at same Load CO2 Stripper Detail Ammonia Stripper unit shell side Tube side shell side Steam Urea- carbamate solution and CO2 Steam Urea-carbamate solution 294066 Kg/Hr 58562 100915 42000 0 C 215.9 185 222 0 C 215.9 176 222 204,211(Megabond) kg/cm2 a 22.8 153 24 kg/cm2 a 26 170 27 0 C 300 230 300 Research Gate is an academic social networking site 8 This is an open access article, Research Gate is a European commercial social networking site for scientists and researchers Ammonia Stripper Tube side carbamate solution and CO2 211885 49589 188 204,211(Megabond) 150 175 235
- 9. www.researchgate.net 28 th Sept, 2022 DOI: 10.13140/RG.2.2.16894.66880 Research Gate is an academic social networking site 9 This is an open access article, Research Gate is a European commercial social networking site for scientists and researchers Mechanical features Parameters Ammonia stripping CO2 Stripping No. of tubes 2600 1677 Tube material X2 25-22-2 (Cr-Ni-Mo-N) Stainless steel Bimetallic, 2RE69+Zr, Mega bond (Ti+ Zr) Tube I.D,mm 25 20 Tube O.D.,mm 31 25.4 Tube length,mm 6000 6000 Tube pitch,mm 41 41 Surface area, m2 1519 632 Shell material carbon steel carbon steel Stripper Material and Heat balance Stripper Inlet stream Stripper Inlet Components Kg/hr K mole/hr mole fraction, x sp heat KJ/K mol0 C,Cp Σ x.cp Ammonia 70350 4138.24 0.51 98.90 50.10 CO2 29200 663.64 0.08 58.96 4.79 Urea 73560 1226.00 0.15 121.32 18.21 Water 38500 2138.89 0.26 74.00 19.38 Biuret 240 2.33 0.0003 183.80 0.05 Air 803 27.69 0.0034 31.61 0.11 Total 212653 8169.09 1.00 568.59 92.63 Heat Input 143776631 KJ/HR Stripper outlet stream (Liquid) Stripper Outlet Liquid Components Kg/hr Wt % mole % Mol Fraction, x Sp. Heat, Cp Kg Mol/Hr Σ x.cp Ammonia 38234 23.46 1.38001081 0.382123939 83.6 2249.06 31.95 CO2 12840 7.88 0.17905812 0.049581057 56.8 291.82 2.82 Urea 73532 45.12 0.75198089 0.208222933 186 1225.53 38.73 Water 38100 23.38 1.29877567 0.359629991 79.9 2116.67 28.73 Biuret 268 0.16 0.00159654 0.00044208 202 2.60 0.09 Total 162974 100 3.61142203 1.00 5885.68 102.31 Heat carried out by stream=5885.68*102.31*205=12.34*107 kJ/Hr
- 10. www.researchgate.net 28 th Sept, 2022 DOI: 10.13140/RG.2.2.16894.66880 Research Gate is an academic social networking site 10 This is an open access article, Research Gate is a European commercial social networking site for scientists and researchers Stripper outlet stream (Vapour) Stripper Outlet Vapour Components Kg/hr Wt % mole % Mol Fraction, x Sp. Heat, Cp Kg Mol/Hr Σ x. cp M *λ Ammonia 34116 68.13% 0.04 0.85 106.76 2006.82 90.96 52879800 CO2 14360 28.68% 0.01 0.14 59.96 326.36 8.31 9334000 Water 400 0.80% 0.0004 0.01 79.80 22.22 0.75 744000 Inerts 1200 2.40% 0.0009 0.02 34.00 42.86 0.62 Total 50076 100.00% 0.05 1.00 246.52 2355.41 100.02 62957800 Sensible Heat of vapour 45940162.51 KJ/Hr Latent Heat of Vaporization 62957800 KJ/Hr Total 108897962.5 KJ/Hr Heat (steam) supplied to stripper in shell side Steam Flow- 50 .0 T/Hr, at 24 ata, 2220 C, Latent heat 1771.4 KJ/Kg Total Heat= 50000 *1771=8.857 *107 KJ/Hr Now Heat input=Heat output 14.37 *107 + 8.857*107 =12.34*107 +10.88*107 23.22*107 KJ/Hr=23.22*107 KJ/Hr. Calculations for estimation of additional energy to be removed to bring down CO2 temperature from 50ºC to 40ºC CO2 flow at the exit of the Separator B-3306 Components Flow (Nm³/h CO2 49349 N2 50 H2 323 H2O 4446 Total flow (dry) 49722 Total flow (wet) 54168 Water (liquid) at the bottom of the separator=15764 kg/hr =15764/18*22.414 Nm³/h=19630 Nm3 /hr Hence total water vapour at the inlet of 1st regeneration overhead condenser=4446+19630-24076 Nm3 /hr. CO2 Flow at inlet of regenerator overhead condenser. Sr. No. Components Flow, Nm3 /hr Mole Fraction Mole fraction value Molecular weight with mole fraction Molecular weight, kg 1 CO2 49349 49349/73798= 0.6687 44*0.6687= 29.42 2 N2 50 50/73798= 0.0007 28*0.0007= 0.0196 3 H2 323 323/73798= 0.0044 2*0.0044= 0.00288 4 H2O 24076 24076/73798= 0.3262 18*0.3262= 5.816
- 11. www.researchgate.net 28 th Sept, 2022 DOI: 10.13140/RG.2.2.16894.66880 Research Gate is an academic social networking site 11 This is an open access article, Research Gate is a European commercial social networking site for scientists and researchers 5 Total (dry) 49722 6 Total (wet) 73798 7 Molecular weight of the Gas 35.32 Quantity of water condensation at 400 C Vapour of water at 400 C=0.073 kg/cm2 a Total Pressure=0.5 kg/cm2 g Hence Absolute pressure=0.5 +0.9928 kg/cm2 a=1.4928 kg/cm2 a Hence water vapour at the exit of the separator at 400 C= 𝟒𝟗𝟕𝟐𝟐∗𝟎.𝟎𝟕𝟑 𝟏.𝟒𝟗𝟐𝟖 𝟎.𝟎𝟕𝟑 =2556 Nm3/hr Hence Total water vapour condensed =24056-2556=21520 Nm3 /hr 𝐇𝐞𝐧𝐜𝐞 𝐓𝐨𝐭𝐚𝐥 𝐰𝐚𝐭𝐞𝐫 𝐯𝐚𝐩𝐨𝐮𝐫 𝐜𝐨𝐧𝐝𝐞𝐧𝐬𝐞𝐝 = 𝟐𝟏𝟓𝟐𝟎∗𝟏𝟖 𝟐𝟐.𝟒 Kg/Hr=17282 kg/hr. Cp of the gas=0.24 kcal Gas inlet temperature to separator=93.90 C Gas outlet temperature to separator=400 C Latent Heat of water at 400 C=577 k.cal/kg Hence the Total Heat load= =73798*0.24*53.9*35/22.414+17282*577 =11462424 k.cal/hr 11.46 G.Cal/hr Heat load of the existing condenser in revamp case=10.24 G.Cal/Hr Hence, requirement of additional heat load=11.46-10.24=1.22 G.Cal/hr Pool Reactor calculation Sr. No Components % Moles 1 Ammonia 29.60 1.741 2 CO2 18.90 0.430 3 Urea(include biuret) 33.00 0.550 4 Water 18.50 1.028 5 Total 100.00 3.748 Sr. No. CO2 required for 0.55 moles of urea 0.550 moles 1 Reactor outlet CO2 is 0.430 moles Therefore Reactor inlet CO2 would be 0.550+0.430=0.980 moles 2 NH3 required for 0.550 moles of Urea 0.550*2=1.10 moles Reactor outlet NH3 is 1.741 moles Therefore Reactor inlet NH3 would be 1.10+1.741=2.841 moles 3 H2O at Reactor inlet 1.028-0.550=0.478 moles 𝑃𝑒𝑟𝑐𝑒𝑛𝑡𝑎𝑔𝑒 𝐶𝑜𝑛𝑣𝑒𝑟𝑠𝑖𝑜𝑛 = Inlet CO2 − Outlet CO2 Inlet CO2 𝑃𝑒𝑟𝑐𝑒𝑛𝑡𝑎𝑔𝑒 𝐶𝑜𝑛𝑣𝑒𝑟𝑠𝑖𝑜𝑛 = . . . ==56.1% N/C Ratio=2.841/0.980=2.9.H/C ratio=0.478/0.980=0.49
- 12. www.researchgate.net 28 th Sept, 2022 DOI: 10.13140/RG.2.2.16894.66880 Research Gate is an academic social networking site 12 This is an open access article, Research Gate is a European commercial social networking site for scientists and researchers Fig- CO2 stripping (small plant 188 TPD) Conclusion The era of small plants has gone, now is the era of big mega urea plants, the stripper has a huge role in that. The plant’s capacity depends upon stripper. Now the era is one ammonia and one urea single stream plants. Earlier there were one ammonia and two urea plants. Now the era is of 4000-5000 TPD urea plants.Big plants mean everything is bigger too much. explosive hazards and more stress on equipments lines. There are many problems in start up and shut down in mega plants. The more passivation air also creates safety issues. Fixing corroded stripper tubes is exceptionally costly. Even a few days of downtime can cost businesses millions in lost revenue. Replacing strippers every 10 years is also a time-consuming and costly procedure. ************************************************************************************